Rhinology
Vol. 46: Issue 2 - April 2026
Dose-dependent IL-29 activation of TLR4 signaling drives eosinophil infiltration in chronic rhinosinusitis with nasal polyps
Summary
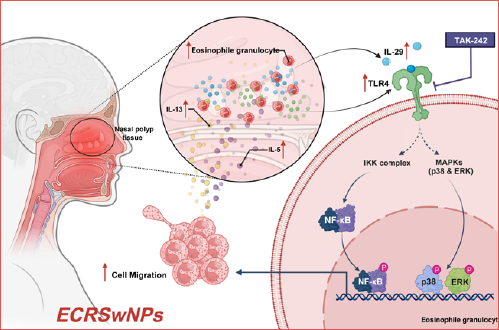
Objective. Eosinophilic chronic rhinosinusitis with nasal polyps (ECRSwNP) features extensive eosinophil infiltration, yet the molecular mechanisms driving this process are not fully elucidated. IL- 29 and TLR4 are known inflammatory modulators, but their dose-dependent interplay in ECRSwNP remains uncharted. This study aimed to explore how IL-29 activates TLR4 signalling to promote eosinophil infiltration in ECRSwNP.
Methods. Thirty patients with ECRSwNP and 30 controls post-nasal septum correction were recruited. Eosinophil infiltration was assessed via haematoxylin-eosin staining, while IL-29 and TLR4 expression and correlation were analysed using qPCR and immunohistochemistry. In vitro, eosinophils were stimulated with IL-29 (10-100 ng/mL) ± TLR4 inhibitor TAK-242, with migration measured by Transwell assay, cytokine secretion by ELISA, and NF-κB/MAPK signalling by western blot.
Results. Patients with ECRSwNP exhibited significantly elevated eosinophil infiltration and IL-29/ TLR4 expression (p < 0.05), with a robust correlation (r = 0.6018, p < 0.0001). IL-29 dose-dependently enhanced eosinophil migration and cytokine production, and the effects were reversed by TLR4 blockade, accompanied by decreased NF-κB and MAPK phosphorylation, indicating TLR4-mediated regulation.
Conclusions. Dose-dependent IL-29 activation of TLR4 signalling drives eosinophil infiltration in ECRSwNP, offering novel mechanistic insights and potential therapeutic targets for this condition.
Introduction
Eosinophilic chronic rhinosinusitis with nasal polyps (ECRSwNP) is a common otolaryngologic disorder characterised by sinus inflammation and nasal polyps formation, accompanied by significant eosinophilic infiltration 1. This condition affects respiratory function and leads to olfactory dysfunction, headaches, and facial pressure, severely impacting patients’ quality of life 2. Globally, the prevalence of ECRSwNP has been increasing annually, making it a public health concern 3. Abnormal activation and aggregation of eosinophils are central features of the pathophysiology of ECRSwNP, yet the precise inflammatory mechanisms remain incompletely understood 4. Identifying and understanding the molecular mechanisms underlying eosinophil activation and recruitment 5 are crucial for developing more effective therapeutic strategies 6,7.
Previous studies identified various cytokines and inflammatory mediators as key players in ECRSwNP, particularly eosinophil-associated cytokines such as IL-5 and IL-13 8,9. In recent years, IL-29 (IFN-λ1) has emerged as a novel immunomodulator implicated in multiple inflammatory diseases. IL-29 has been shown to regulate immune cell functions and participate in inflammatory responses 10. Also, toll-like receptor 4 (TLR4), a key component of the innate immune system, has been extensively studied for its role in infectious and non-infectious inflammation. TLR4 activation triggers a cascade of signalling events 11-13, leading to the production and release of inflammatory cytokines 14. However, the roles of IL-29 and TLR4 in ECRSwNP and their potential interactions remain largely unexplored, limiting a comprehensive understanding of the disease’s complex pathophysiology.
Although the independent roles of IL-29 and TLR4 have been well characterised in other inflammatory diseases, studies investigating their expression patterns, interactions, and specific effects on eosinophil function in ECRSwNP are scarce 15. Notably, there is a lack of systematic research assessing whether IL-29 and TLR4 expression levels correlate with ECRSwNP severity and whether they regulate eosinophil activity and inflammatory mediator secretion through shared or distinct pathways. Furthermore, the precise role of the TLR4 signalling pathway in IL-29-induced eosinophil activation remains unclear 16. Elucidating these mechanisms is critical for uncovering the molecular basis of the disease and developing novel therapeutic strategies.
This study aims to fill this knowledge gap by comprehensively analysing the expression levels and correlation of IL-29 and TLR4 in ECRSwNP patients and investigating their joint regulatory effects on eosinophil function. A comparative study has been conducted between ECRSwNP and non-ECRSwNP patients, utilising quantitative PCR (qPCR) and immunohistochemistry (IHC) to quantify IL-29 and TLR4 expression levels. Additionally, in vitro experiments have been performed to evaluate the effects of IL-29 on eosinophil migration and inflammatory cytokine secretion. The role of the TLR4 signalling pathway in mediating IL-29-induced effects was further examined using the TLR4 inhibitor TAK-242. The findings of this study are expected to enhance our understanding of the pathophysiological mechanisms underlying ECRSwNP and provide novel molecular targets for its diagnosis and treatment. These insights may contribute to developing targeted therapeutic strategies, ultimately improving patient outcomes and quality of life.
Materials and methods
Study participants
This study enrolled a total of 60 patients who were hospitalised in the Department of Otorhinolaryngology-Head and Neck Surgery at Hunan Provincial People’s Hospital and underwent functional endoscopic sinus surgery (FESS) or septoplasty between November 2021 and December 2022. The inclusion criteria were as follows: age ≥ 18 years, with the experimental group consisting of 30 patients diagnosed with ECRSwNP, defined by postoperative pathological results showing an eosinophil infiltration ratio of ≥ 10%. The control group included 30 healthy individuals undergoing septoplasty due to congenital anatomical abnormalities of the nasal septum, with no history of chronic rhinosinusitis. Detailed information is provided in Figure S1. All patients provided written informed consent before surgery.
The following exclusion criteria were applied: patients with comorbid asthma, chronic obstructive pulmonary disease (COPD), or allergic rhinitis; patients diagnosed with nasal malignancies, autoimmune diseases, or immunodeficiency disorders; and those who had used glucocorticoids, steroid medications, or antihistamines within one month before surgery.
Sample collection
Tissue samples from both the experimental and control groups were collected during surgery. Nasal polyp tissue samples were obtained intraoperatively for patients in the experimental group. The samples were continuously rinsed with physiological saline to remove all surface blood and mucus secretions, then blotted dry using sterile gauze. Each sample was divided into 2 parts: one was fixed in formalin for pathological examination, while the other was immediately preserved in liquid nitrogen and transferred to a -80°C freezer for storage. For the control group, tissue samples were collected from the inferior turbinate mucosa. After thorough washing, these samples were also stored at -80°C for subsequent analysis.
Eosinophil isolation and culture
Peripheral blood from healthy volunteers was collected, and eosinophils were isolated using Ficoll-Paque density gradient centrifugation (GE Healthcare, 17-1440-02). The purified cells were cultured in RPMI-1640 medium (Gibco, 31800-022) supplemented with 10% fetal bovine serum (Gibco, 10270-106) and 1% penicillin-streptomycin (Gibco, 15140-122) at 37°C in a 5% CO2 incubator. The cells were used for subsequent functional assays, and the experimental period did not exceed 48 hours to ensure cell viability.
IL-29 dose-dependent stimulation assay
To investigate the dose-dependent effects of IL-29 on eosinophil function, cells were divided into a control group (untreated) and IL-29 stimulation groups with different concentrations (10, 25, 50, and 100 ng/mL, R&D Systems, 1598-IL-025/CF). IL-29 was added to the culture medium, and after 24 hours of stimulation, cells and supernatants were collected for functional assays and signalling pathway analysis.
TLR4 blockade assay
To evaluate the role of TLR4 in IL-29-mediated eosinophil regulation, a TLR4 blockade group (IL-29 50 ng/mL + TAK-242 10 μM, MedChemExpress, HY-11109) and a TAK-242-only intervention group were established. TAK-242 was pre-incubated with cells for one hour before the stimulation of IL-29 (10, 25, 50, 100 ng/mL). Cells were then co-stimulated for 24 hours, and eosinophil migration, inflammatory cytokine secretion, and signalling pathway activation were assessed.
Cell migration assay
Eosinophil migration ability was assessed using Transwell chambers (Corning, 3422). A total of 1×105 eosinophils were seeded in the upper chamber, while the lower chamber was filled with RPMI-1640 medium containing 10 ng/mL IL-5 (R&D Systems, 205-IL-010) as a chemoattractant. After incubation for 4 hours, migrated cells on the lower membrane surface were fixed with 4% paraformaldehyde (Sigma, F8775) and stained with 0.1% brilliant green (Solarbio, G1014). The number of transmigrated cells was counted under a microscope to compare migration capacity among different groups.
Inflammatory cytokine secretion assay
The levels of IL-5 and IL-13 in the cell culture supernatants were measured using commercial ELISA kits (Thermo Fisher Scientific, BMS278 and BMS231-3). The assay was performed according to the manufacturer’s instructions. Optical density (OD) values were recorded using a microplate reader (Thermo Fisher Scientific, Multiskan FC), and cytokine concentrations were calculated accordingly.
Western blot analysis
Total eosinophil proteins were extracted using RIPA lysis buffer (Beyotime, P0013B), and protein concentration was determined using the BCA protein quantification kit (Beyotime, P0012). The total protein was separated by SDS-PAGE electrophoresis, transferred onto a membrane, and incubated with primary antibodies against TLR4 (Abcam, ab217274), phosphorylated NF-κB (p-NF-κB) (CST, 3033S), phosphorylated MAPK (p-MAPK) (CST, 4370S), and β-actin (CST, 4970S) as an internal control. After incubation with HRP-conjugated secondary antibodies (CST, 7074S), protein bands were detected using enhanced chemiluminescence (ECL) (Thermo Fisher Scientific, 32106). Band intensity was quantified using ImageJ software.
IHC method
Tissue samples were fixed in 4% paraformaldehyde (Sigma, F8775), followed by routine dehydration, embedding, and sectioning at a thickness of 4 μm. After deparaffinisation and rehydration through a graded ethanol series, antigen retrieval was performed using 10 mM sodium citrate buffer (pH 6.0, Beyotime, P0081), heated in a 95°C water bath for 10 minutes, and then cooled to room temperature. Endogenous peroxidase activity was blocked by incubating the sections with 3% H2O2 (Beyotime, ST038) for 10 minutes, followed by blocking with 5% bovine serum albumin (BSA, Beyotime, A8020) at room temperature for 30 minutes.
The sections were incubated overnight at 4°C with primary antibodies against IL-29 (Abcam, ab244788, 1:200 dilution) and TLR4 (CST, 14358S, 1:300 dilution). The following day, the sections were washed 3 times with PBS (5 minutes each) and then incubated with HRP-conjugated secondary antibody (Beyotime, A0208, 1:500 dilution) at room temperature for one hour, followed by another 3 washes with PBS. DAB chromogenic reagent (Solarbio, DA1010) was used for staining, with the colour development time controlled under a microscope (approximately 1-3 minutes). After staining, the sections were counterstained with haematoxylin (Solarbio, G1140), dehydrated through a graded ethanol series, cleared with xylene, and mounted using neutral resin (Beyotime, G8590).
Staining results were observed under a Nikon ECLIPSE Ci microscope, with positive signals appearing as brownish-yellow staining in the cytoplasm or on the cell membrane. Five random high-power fields (400× magnification) were selected per section for imaging. ImageJ software quantified the proportion of positive cells and staining intensity scores (Immunoreactive Score, IRS).
Data analysis
All statistical analyses and figure generation were conducted using SPSS 27.0 and GraphPad Prism 8.0. Measurement data were first assessed for normality and homogeneity of variance. Results were expressed as mean ± standard deviation (SD) for data that followed a normal distribution, and ANOVA or t-tests were used for group comparisons. If the data did not meet the normality assumption, results were presented as median with interquartile range (IQR) and analysed using non-parametric tests. Categorical data were described using frequencies and percentages, with group comparisons performed using the χ2 (chi-square) test; when the expected frequency was < 5, Fisher’s exact test was used. Pearson correlation analysis was applied for correlation assessments. Statistical significance was set at α = 0.05, with p < 0.05 considered statistically significant.
Results
Expression and correlation analysis of IL-29 and TLR4 in ECRSwNP
No significant differences were observed in the clinical characteristics between the experimental group (nasal polyps with ≥ 10% eosinophil infiltration) and the control group (patients undergoing septoplasty with a history of chronic rhinosinusitis), ensuring the comparability of the 2 groups. We further investigated the expression characteristics and correlation of IL-29 and TLR4 in ECRSwNP through histological and molecular biology analyses.
Haematoxylin-eosin staining revealed that eosinophils in the experimental group were round or oval, with mononuclear or lobulated nuclei, staining bright red or pink. In all 30 patients from the experimental group, significant eosinophil infiltration was observed under the microscope, accompanied by tissue oedema and glandular hyperplasia, whereas no or minimal eosinophil infiltration was detected in the control group (Fig. 1A).
qPCR analysis further demonstrated that the mRNA expression levels of IL-29 and TLR4 in the nasal polyp tissues of the experimental group were significantly higher than those in the control group (p < 0.0001) (Fig. 1B). Correlation analysis indicated a positive correlation between IL-29 and TLR4 in the experimental group (r = 0.6018, p < 0.0001) (Fig. 1C).
IHC results showed that both IL-29 and TLR4 were expressed in the experimental and control groups; however, the positive expression rate in the experimental group was significantly higher than in the control group (p < 0.05). Specifically, the positive rate of IL-29 in the experimental group was 76.6%, while that of TLR4 was 80%. Additionally, the IRS for both IL-29 and TLR4 was significantly higher in the experimental group compared to the control group, with statistically significant differences (Figs. 1D,E) (Tabs. I-III). Furthermore, cell-specific staining patterns revealed that IL-29 was predominantly localised in the cytoplasm of respiratory epithelial cells, including goblet and basal cells. In contrast, TLR4 expression was observed both in epithelial cells and in submucosal inflammatory infiltrates, especially in eosinophil-like granulocytes.
IL-29 enhances eosinophil migration
Eosinophil migration was significantly influenced by IL-29 stimulation, exhibiting an apparent dose-dependent effect (Fig. 2). At a low concentration (10 ng/mL), the number of migrated cells slightly increased compared to the control group. However, in the moderate (25 ng/mL) and high concentration (50 ng/mL) groups, migration capacity was markedly enhanced, with the 50 ng/mL group reaching its peak. In contrast, the exceptionally high concentration (100 ng/mL) group did not further enhance migration ability, suggesting that the promotive effect of IL-29 on migration becomes saturated at higher concentrations (Figs. 2A,B). These findings indicate that IL-29 effectively promotes eosinophil migration in a dose-dependent and saturable manner.
IL-29 promotes eosinophil secretion of inflammatory cytokines
IL-29 stimulation also significantly affected inflammatory cytokine secretion, demonstrating a dose-dependent pattern (Fig. 3). Upon IL-29 stimulation, the secretion of IL-5 and IL-13 by eosinophils was markedly increased. The low-concentration group (10 ng/mL) exhibited slightly higher cytokine levels compared to the control group, whereas the moderate (25 ng/mL) and high-concentration (50 ng/mL) groups showed a substantial increase in cytokine secretion. In the high concentration group, IL-5 and IL-13 secretion levels reached approximately 3-fold and 2.5-fold those of the control group, respectively, indicating a strong promotive effect of IL-29 on inflammatory cytokine secretion (Fig. 3A,B). Similar to the migration results, the very high concentration (100 ng/mL) group did not further enhance cytokine secretion, reinforcing the saturation effect of IL-29.
IL-29 activates the TLR4 downstream signaling pathway
To further explore the mechanism underlying IL-29 function, we examined the activation of TLR4 and its downstream signalling molecules p-NF-κB and p-MAPK (Fig. 4). Western blot analysis demonstrated that IL-29 stimulation significantly increased TLR4 protein expression dose-dependently. In the low-concentration group (10 ng/mL), the activation levels of TLR4 and p-NF-κB were slightly higher than those in the control group. However, in the moderate (25 ng/mL) and high concentration (50 ng/mL) groups, TLR4 activation (Fig. 4A), p-NF-κB phosphorylation (Fig. 4B), and p-MAPK phosphorylation (Fig. 4C) were markedly upregulated. Notably, in the very high concentration group (100 ng/mL), the activation of the signaling pathway did not further increase, supporting the conclusion that IL-29 exhibits a dose-dependent and saturable effect.
TAK-242 blocks TLR4 and inhibits IL-29 effects
To determine whether the effects of IL-29 depend on the TLR4 signalling pathway, functional experiments were conducted with the TLR4 inhibitor TAK-242 (Fig. 5). Blocking TLR4 significantly suppressed IL-29-induced eosinophil migration and inflammatory cytokine secretion. Transwell migration assays demonstrated that regardless of IL-29 concentration, the presence of TAK-242 significantly reduced migration capacity (Fig. 5A). Furthermore, ELISA revealed that TAK-242 significantly inhibited the secretion of IL-5 and IL-13 (Fig. 5B,C). Western blot analysis further confirmed that TAK-242 markedly decreased IL-29-induced expression of TLR4, p-NF-κB, and p-MAPK (Fig. 5D).
These findings collectively indicate that IL-29’s effects on eosinophil migration and inflammatory cytokine secretion are dependent on activation of the TLR4 signalling pathway.
Discussion
This study investigated the expression and synergistic effects of IL-29 and TLR4 in ECRSwNP, revealing their potential pro-inflammatory mechanisms. ECRSwNP are characterised by eosinophilic infiltration and a T2-type inflammatory response, but the precise regulatory mechanisms remain unclear. Previous studies have indicated that IL-29 plays a significant role in viral infections, autoimmune diseases, and chronic inflammation 17,18; however, its role in ECRSwNP has not been well defined. This study found that IL-29 and TLR4 were significantly upregulated and positively correlated in ECRSwNP tissues (r = 0.6018, p < 0.0001). Further in vitro experiments demonstrated that IL-29 promotes eosinophil migration and induces IL-5 and IL-13 secretion, with its effects dependent on TLR4-mediated signalling pathways. These findings provide novel insights into the inflammatory mechanisms of ECRSwNP and suggest that the IL-29/TLR4 axis may serve as a potential therapeutic target.
IL-29, also known as interferon-λ1, is a type III interferon strongly induced by viral infections, especially in epithelial tissues 19,20. While originally characterised by its antiviral effects, IL-29 has also been shown to regulate immune polarisation and epithelial-immune interactions, potentially contributing to T2-type inflammation in certain contexts 21. In ECRSwNP, recurrent viral exposure may lead to sustained IL-29 expression, which in turn can activate TLR4-dependent eosinophilic responses, as demonstrated in this study. These findings provide a plausible mechanistic link between viral triggers and chronic T2-skewed inflammation via the IL-29/TLR4 axis. Further studies using viral infection models of nasal polyps are warranted to validate this potential pathway.
The role of IL-29 in chronic inflammatory diseases remains controversial. Studies have shown that IL-29 primarily regulates immune cell function through the JAK-STAT signalling pathway, exhibiting either pro-inflammatory or anti-inflammatory effects in autoimmune diseases such as systemic lupus erythematosus and rheumatoid arthritis 22. Our study found that IL-29 is significantly upregulated in ECRSwNP tissues and correlates with eosinophilic infiltration, suggesting its role in promoting T2-type inflammation. Additionally, in vitro experiments demonstrated that IL-29 enhances eosinophil migration and IL-5/IL-13 secretion in a dose-dependent manner. Previous studies have also reported that IL-29 induces IL-25 and IL-33 release in airway epithelial cells, exacerbating allergic inflammation 23,21. Therefore, the role of IL-29 in eosinophilic inflammation associated with ECRSwNP warrants further investigation to determine whether it could serve as a biomarker or therapeutic target for the disease.
TLR4 has been widely recognised as a key regulator in multiple chronic inflammatory diseases 24. Previous studies have demonstrated that TLR4 activation can trigger NF-κB and MAPK signalling pathways, leading to the secretion of inflammatory cytokines. Our study revealed that TLR4 expression was significantly higher in ECRSwNP tissues than in the control group, and it was positively correlated with IL-29 expression, suggesting that IL-29 may promote ECRSwNP-related inflammation through the TLR4-mediated signalling pathway. Further in vitro experiments confirmed that IL-29 activates p-NF-κB and p-MAPK signalling in eosinophils, enhancing cell migration and cytokine secretion. Moreover, blocking TLR4 significantly attenuated IL-29-induced pro-inflammatory effects, indicating that TLR4 plays a crucial role in IL-29-mediated eosinophil activation. These findings deepen our understanding of the immunological mechanisms underlying ECRSwNP and provide experimental evidence supporting TLR4 as a potential therapeutic target.
Beyond their classical role in promoting T2-type inflammation through cytokine secretion, eosinophils also actively participate in local tissue remodelling and epithelial barrier disruption in ECRSwNP. Upon activation, eosinophils release cytotoxic granule proteins, such as major basic protein and eosinophil cationic protein, which directly damage the epithelium and contribute to mucosal remodelling and glandular hyperplasia 25,26. Furthermore, eosinophils interact with epithelial and stromal cells, amplifying local inflammatory loops and altering the tissue microenvironment 27. The sustained presence of activated eosinophils perpetuates chronic inflammation and drives polyp formation 28. These local effects contextualise the functional findings observed in this study and highlight eosinophils as critical effector cells. Understanding how IL-29/TLR4 signalling modulates these local actions may reveal novel therapeutic avenues.
To further verify the critical role of TLR4 in IL-29-mediated pro-inflammatory effects, this study employed the TLR4 inhibitor TAK-242 for intervention. The results demonstrated that TAK-242 significantly inhibited IL-29-induced eosinophil migration and the secretion of IL-5 and IL-13, reducing the phosphorylation levels of p-NF-κB and p-MAPK. These findings indicate that IL-29 exerts its pro-inflammatory effects through the TLR4-mediated signalling pathway and that TAK-242 may serve as a potential therapeutic agent for modulating inflammation in ECRSwNP. Previous studies have demonstrated that TAK-242 exhibits potent anti-inflammatory effects in diseases such as rheumatoid arthritis, inflammatory bowel disease, and asthma 29, providing a reference for its further exploration in the field of ECRSwNP. Future research should further evaluate the clinical therapeutic potential of TAK-242 in treatment of ECRSwNP and investigate whether it can alleviate symptoms and reduce eosinophilic inflammation in patients.
Compared with previous studies, this study is the first to reveal the synergistic role of IL-29 and TLR4 in ECRSwNP tissues and to validate their critical function in eosinophil activation through in vitro experiments. Prior research has primarily focused on the role of TLR4 in nasal mucosal epithelial cells or macrophages, whereas this study expands its known function to eosinophils, thereby enriching the immuno-inflammatory research on ECRSwNP. Furthermore, this study identifies TLR4 as a mediator of IL-29-induced pro-inflammatory effects, a mechanism that has not been previously reported in ECRSwNP research. These findings provide new theoretical insights into the immunoregulatory mechanisms of this disease but also offer experimental evidence supporting the IL-29/TLR4 axis as a potential therapeutic target.
This study, at both the molecular and cellular levels, elucidates the key roles of IL-29 and TLR4 in ECRSwNP and clarifies their synergistic regulation of eosinophil function. These findings provide new scientific evidence for understanding the inflammatory mechanisms of ECRSwNP. Clinically, IL-29 and TLR4 may serve as potential biomarkers for ECRSwNP, aiding in early diagnosis and disease assessment. Furthermore, targeting the IL-29/TLR4 signalling pathway, particularly with TLR4 inhibitors (such as TAK-242), may offer novel therapeutic strategies for ECRSwNP with significant translational potential.
Although this study reveals the inflammatory regulatory roles of IL-29 and TLR4 through histological analysis and in vitro experiments, certain limitations remain. First, this study primarily focuses on gene and protein expression levels and in vitro functional assays, without in vivo validation using animal models. Additionally, the direct interaction between IL-29 and TLR4 has not been comprehensively explored through advanced molecular techniques, such as co-immunoprecipitation or single-cell multi-omics analysis. Lastly, the sample size is limited, and different pathological subtypes of ECRSwNP were not included, which may impact the generalisability of the findings.
Future research should further explore the roles of IL-29 and TLR4 in different pathological subtypes of ECRSwNP and expand the sample size to enhance the reliability and generalisability of the findings. Additionally, in vivo studies using animal models should be conducted to systematically evaluate the functions of IL-29 and TLR4 and their dynamic changes in disease progression. Furthermore, single-cell multi-omics technologies should be utilised to elucidate the cell-specific mechanisms of IL-29 and TLR4, and more efforts should be devoted to exploring precision-targeted intervention strategies against TLR4 or IL-29. These investigations will provide a more comprehensive theoretical foundation and technical support for diagnosing and treating ECRSwNP.
Conclusions
This study presents the comprehensive demonstration that IL-29 promotes eosinophil migration and inflammatory cytokine secretion in ECRSwNP via TLR4-dependent signaling. The upregulated and positively correlated expression of IL-29 and TLR4 in patient tissues, together with in vitro mechanistic evidence, underscores the IL-29/TLR4 axis as a novel pro-inflammatory pathway. These findings not only enrich our understanding of the immunopathogenesis of ECRSwNP but also suggest IL-29 and TLR4 as promising biomarkers and therapeutic targets. Targeted modulation of this signaling cascade – such as through TLR4 inhibition with TAK-242 – may provide a new avenue for personalised treatment. Future studies are warranted to validate these mechanisms in vivo, explore their role in different disease subtypes, and investigate the translational potential of TLR4-targeted therapies.
Conflict of interest statement
The authors declare no conflict of interest.
Funding
This study was supported by Provincial Natural Science Foundation Project (No.2025JJ80748).
Author contributions
YZ, JL: conceived and designed the study; HY, YZ, NH: performed the experiments; YRZ, NH: analyzed the data; YZ, JL: wrote the manuscript. All authors reviewed and approved the final version of the manuscript.
Ethical consideration
This study was reviewed and approved by the Ethics Committee of Hunan Provincial People’s Hospital (Approval Number: 2023-143). All procedures involving human participants were conducted in accordance with the ethical standards of the institutional research committee and the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all participants prior to their inclusion in the study, with participants fully informed of the study’s objectives, procedures, and their right to withdraw at any time without consequence.
History
Received: April 9, 2025
Accepted: June 10, 2025
Figures and tables
Figure 1. Histological and molecular expression analysis of IL-29 and TLR4 in ECRSwNP. (A) Haematoxylin-eosin staining of the experimental and control groups; magnification 100×; (B) qPCR analysis of IL-29 and TLR4 mRNA expression and their correlation; (C) Correlation analysis of IL-29 and TLR4, showing a positive correlation in the experimental group (r = 0.6018, p < 0.0001); (D) Immunohistochemical expression of IL-29 in the experimental and control groups (D1: control group, 100×; D2: experimental group, 100×; D3: control group, 400×; D4: experimental group, 400×); (E) Immunohistochemical expression of TLR4 in the experimental and control groups (E1: control group, 100×; E2: experimental group, 100×; E3: control group, 400×; E4: experimental group, 400×). qPCR data are presented as mean ± SD, correlation analysis was performed using Pearson correlation coefficient, and p < 0.05 was considered statistically significant. Experimental group n = 30, control group n = 30.
Figure 2. Effect of IL-29 on eosinophil migration capacity. (A) Transwell assay for eosinophil migration; (B) Statistical analysis of eosinophil migration under different IL-29 concentrations (10, 25, 50, 100 ng/mL). Data were obtained from 3 independent experiments and are presented as mean ± SD. One-way ANOVA was used for statistical analysis. * indicates p < 0.05.
Figure 3. Effect of IL-29 on eosinophil inflammatory cytokine secretion. (A) ELISA analysis of IL-5 secretion in eosinophil supernatants; (B) ELISA analysis of IL-13 secretion in eosinophil supernatants. Eosinophils were stimulated with different concentrations of IL-29 (10, 25, 50, 100 ng/mL) for 24 hours. Data were obtained from 3 independent experiments and are presented as mean ± SD. * indicates p < 0.05.
Figure 4. Effect of IL-29 on TLR4 and downstream signaling molecule activation. (A) Western blot analysis of TLR4 protein expression; (B) Western blot analysis of p-NF-κB protein expression; (C) Western blot analysis of p-MAPK protein expression. Eosinophils were stimulated with different concentrations of IL-29 (10, 25, 50, 100 ng/mL) for 24 hours. β-actin was used as the internal control. Data were obtained from 3 independent experiments. * indicates p < 0.05.
Figure 5. The interventional effect of TAK-242 Blocking TLR4 on IL-29 action. (A) Transwell assay showing changes in eosinophil migration after TLR4 blockade with TAK-242; (B) ELISA of IL-5 secretion after TAK-242 intervention; (C) ELISA of IL-13 secretion after TAK-242 intervention; (D) Western blot analysis of TLR4, p-NF-κB, and p-MAPK protein expression after TAK-242 treatment. Data are presented as mean ± SD. * indicates p < 0.05.
Figure S1. Flowchart of inclusion and exclusion criteria.
| Group | Tissue specimen count | IL-29 Expression Levels | |||||
|---|---|---|---|---|---|---|---|
| Negative (-) | Weakly positive (+) | Positive (++) | Strongly positive (+++) | Positive rate(%) | p value | ||
| Patients | 30 | 16 | 7 | 6 | 1 | 46.6 | 0.017 |
| Controls | 30 | 7 | 13 | 4 | 6 | 76.6* | |
| Comparison with normal nasal mucosal epithelial tissues, ****p < 0.0001; ***p < 0.001; **p < 0.01; *p < 0.05. | |||||||
| Group | Tissue specimen count | TLR4 Expression Levels | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Negative (-) | Weakly positive (+) | Positive (++) | Strongly positive (+++) | Positive rate(%) | p value | |||||
| Patients | 30 | 15 | 13 | 2 | 0 | 50 | 0.015 | |||
| Controls | 30 | 6 | 19 | 3 | 2 | 80* | ||||
| Comparison with normal nasal mucosal epithelial tissues, ****p < 0.0001; ***p < 0.001; **p < 0.01. | ||||||||||
| TLR4 | IL-29 | Total | F | p | |
|---|---|---|---|---|---|
| Positive | Negative | ||||
| Positive | 21 | 3 | 24 | 6.873 | 0.016 |
| Negative | 2 | 4 | 6 | ||
| Total | 23 | 7 | 30 | ||
References
- Brescia G, Contro G, Giacomelli L. Blood eosinophilic and basophilic trends in recurring and non-recurring eosinophilic rhinosinusitis with nasal polyps. Am J Rhinol Allergy. 2020;35:296-301. doi:https://doi.org/10.1177/1945892420953960
- Du K, Pang Z, Lou H. Blood eosinophilia and a higher ethmoid sinus/maxillary sinus score ratio predict new-onset asthma in patients with chronic rhinosinusitis with nasal polyps. Acta Otolaryngol. 2024;144:313-319. doi:https://doi.org/10.1080/00016489.2024.2362776
- Yuan H, Yang Y, Zhang B. Construction and analysis of risk prediction model of eosinophilic chronic rhinosinusitis with nasal polyps: a cross-sectional study in Northwest China. Clin Otolaryngol. 2024;50:39-45. doi:https://doi.org/10.1111/coa.14225
- Long S, Wang H, Li W. Advancements and future directions in chronic rhinosinusitis: understanding inflammatory mechanisms (2000-2023). Eur Arch Otorhinolaryngol. 2025;282:2899-2909. doi:https://doi.org/10.1007/s00405-025-09245-8
- Moreno-Jiménez E, Morgado N, Gómez-García M. TSLP and TSLPR expression levels in peripheral blood as potential biomarkers in patients with chronic rhinosinusitis with nasal polyps. IJMS. 2025;26. doi:https://doi.org/10.3390/ijms26031227
- Zheng M, Wu D, Piao Y. Efficacy and safety of GR1802 in uncontrolled chronic rhinosinusitis with nasal polyps: placebo-controlled phase 2 trial. J Allergy Clin Immunol Pract. 2025;155:1575-1583. doi:https://doi.org/10.1016/j.jaci.2025.01.034
- Caminati M, Maule M, Bagnasco D. Profiling blood hypereosinophilia in patients on dupilumab treatment for respiratory conditions: a real-life snapshot. J Allergy Clin Immunol Pract. 13:1367-1374.e4. doi:https://doi.org/10.1016/j.jaip.2025.01.040
- Wu P, Huang C, Chang P. Blood eosinophil count is the dominant clinical marker for type 2 inflammatory severity in CRSwNP. Laryngoscope. 2024;135:1326-1334. doi:https://doi.org/10.1002/lary.31899
- Lombardo N, D’Ecclesia A, Chiarella E. Real-world evaluation of dupilumab in the long-term management of eosinophilic chronic rhinosinusitis with nasal polyps: a focus on IL-4 and IL-13 receptor blockade. Medicina. 2024;60. doi:https://doi.org/10.3390/medicina60121996
- He S, Li T, Chen H. CD14+ cell-derived IL-29 modulates proinflammatory cytokine production in patients with allergic airway inflammation. Allergy. 2010;66:238-246. doi:https://doi.org/10.1111/j.1398-9995.2010.02455.x
- Lee S, Choi M, Chung J. Povidone iodine suppresses LPS-induced inflammation by inhibiting TLR4/MyD88 formation in airway epithelial cells. Sci Rep. 2022;12. doi:https://doi.org/10.1038/s41598-022-07803-2
- Wang L, Zhao Y, Yao S. Vitamin D improves the effect of glucocorticoids on attenuating lipopolysaccharide-induced IL-6 production via TLR4/NF-κB pathway in human respiratory epithelial cells. Int Arch Allergy Immunol. 2022;183:1017-1028. doi:https://doi.org/10.1159/000524855
- Khayer N, Jalessi M, Farhadi M. S100a9 might act as a modulator of the toll-like receptor 4 transduction pathway in chronic rhinosinusitis with nasal polyps. Sci Rep. 2024;14. doi:https://doi.org/10.1038/s41598-024-60205-4
- Kang J, Yang H, Park J. Lipopolysaccharide regulates thymic stromal lymphopoietin expression via TLR4/MAPK/Akt/NF-κB-signaling pathways in nasal fibroblasts: differential inhibitory effects of macrolide and corticosteroid. Int Forum Allergy Rhinol. 2020;11:144-152. doi:https://doi.org/10.1002/alr.22641
- Xu L, Feng X, Tan W. IL-29 enhances Toll-like receptor-mediated IL-6 and IL-8 production by the synovial fibroblasts from rheumatoid arthritis patients. Arthritis Res Ther. 2013;15. doi:https://doi.org/10.1186/ar4357
- Shi J, Dai L, Gu J. Luteolin alleviates olfactory dysfunction in eosinophilic chronic rhinosinusitis through modulation of the TLR4/NF-κB signaling pathway. Int Immunopharmacol. 2025;148. doi:https://doi.org/10.1016/j.intimp.2025.114189
- Henden A, Koyama M, Robb R. IFN-λ therapy prevents severe gastrointestinal graft-versus-host disease. Blood. 2021;138:722-737. doi:https://doi.org/10.1182/blood.2020006375
- Bagheri-Hosseinabadi Z, Kaeidi A, Rezvani M. Evaluation of the serum levels of CCL2, CCL3, and IL-29 after first and second administrations of the COVID-19 vaccine (Oxford-AstraZeneca®). Immunobiology. 2024;229. doi:https://doi.org/10.1016/j.imbio.2024.152789
- Li M, Liu X, Zhou Y. Interferon-λs: the modulators of antivirus, antitumor, and immune responses. J Leukocyte Biol. 2009;86:23-32. doi:https://doi.org/10.1189/jlb.1208761
- Villenave R, Broadbent L, Douglas I. Induction and antagonism of antiviral responses in respiratory syncytial virus-infected pediatric airway epithelium. J Virol. 2015;89:12309-12318. doi:https://doi.org/10.1128/jvi.02119-15
- Deschildre A, Pichavant M, Engelmann I. Virus-triggered exacerbation in allergic asthmatic children: neutrophilic airway inflammation and alteration of virus sensors characterize a subgroup of patients. Respir Res. 2017;18. doi:https://doi.org/10.1186/s12931-017-0672-0
- Hao N, Zhou Z, Zhang F. Interleukin-29 accelerates vascular calcification via JAK2/STAT3/BMP2 signaling. JAHA. 2023;12. doi:https://doi.org/10.1161/jaha.122.027222
- Li Y, Gao Q, Yuan X. Adenovirus expressing IFN-λ1 (IL-29) attenuates allergic airway inflammation and airway hyperreactivity in experimental asthma. Int Immunopharmacol. 2014;21:156-162. doi:https://doi.org/10.1016/j.intimp.2014.04.022
- Long L, Dai X, Yao T. Mefunidone alleviates silica-induced inflammation and fibrosis by inhibiting the TLR4-NF-κB/MAPK pathway and attenuating pyroptosis in murine macrophages. Biomed Pharmacother. 2024;178. doi:https://doi.org/10.1016/j.biopha.2024.117216
- Carvalho N, Carolino E, Coelho H. Eosinophil granule proteins involvement in acute appendicitis – An allergic disease?. IJMS. 2023;24. doi:https://doi.org/10.3390/ijms24109091
- Radajewski K, Kalińczak-Górna P, Zdrenka M. Short term pre-operative oral corticosteroids – Tissue remodeling in chronic rhinosinusitis with nasal polyps. JCM. 2021;10. doi:https://doi.org/10.3390/jcm10153346
- Sastre B, Rodrigo-Muñoz J, Garcia-Sanchez D. Eosinophils: old players in a new game. J Investig Allergol Clin Immunol. 2018;28:289-304. doi:https://doi.org/10.18176/jiaci.0295
- Pawankar R, Nonaka M. Inflammatory mechanisms and remodeling in chronic rhinosinusitis and nasal polyps. Curr Allergy Asthma Rep. 2007;7:202-208. doi:https://doi.org/10.1007/s11882-007-0073-4
- Mahish C, De S, Chatterjee S. TLR4 is one of the receptors for Chikungunya virus envelope protein E2 and regulates virus induced pro-inflammatory responses in host macrophages. Front Immunol. 2023;14. doi:https://doi.org/10.3389/fimmu.2023.1139808
Downloads
License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright
Copyright (c) 2026 Società Italiana di Otorinolaringoiatria e chirurgia cervico facciale
How to Cite
- Abstract viewed - 358 times
- PDF downloaded - 90 times



