Reviews
Vol. 46: Issue 2 - April 2026
Sinonasal giant cell tumour. Report of four cases and review of the literature
Summary
Objective. Sinonasal giant cell tumour (SnGCT) is very rarely reported. The aim of the study is to present a series of SnGCT treated with an endoscopic or combined approach and to carry out a review of the literature on this topic.
Methods. This case series encompassed the clinical data of all the SnGCT surgically treated with curative intent at two different referral centres, between 2001 and 2024. The information was extrapolated to report epidemiologic and clinical data, surgical approach and follow up. A comprehensive clinical review of the literature was also carried out.
Results. Among 4 patients included, 3 were treated with a pure endoscopic resection and remained disease-free at the last follow up, while one was managed with a combined approach, who is alive with disease. From the review we found only 32 cases of SnGCT reported, mostly treated with an open approach. Local recurrence was documented in 12.5% of cases, while distant metastasis in 3%.
Conclusions. SnGTC is a benign but locally aggressive neoplasm that can present as a lytic lesion. The clinical behaviour of GCT is unpredictable, and it tends to recur locally and may even metastasise. In the nose and paranasal sinuses it is an exceedingly rare entity, but should be considered in the differential diagnosis with a large variety of histologies. Surgery is the treatment of choice for resectable lesions.
Introduction
Sinonasal giant cell tumour (SnGCT) is a benign primary tumour of the bone that represents up to 5% of skeletal neoplasia 1-3. It is most commonly reported in adults, typically affecting the epiphysis of long bones, such as distal femur and proximal tibia, and is slightly more common in females 4. This type of bone tumour is exceedingly rare in head and neck region, accounting for about 2% of all GCT, the majority found in the sphenoid, ethmoid, and temporal bones 5. Nevertheless, it presents certain features that make it relevant in the differential diagnosis of sinonasal masses. Indeed, SnGCT can be misdiagnosed both as fibro-osseous lesions and hypervascularised neoplasms, with biopsy often contraindicated. Moreover, it can be locally aggressive, leading to cortical bone destruction 6,7, with eventually recurrence 8, and some reported cases of malignant transformation and distant metastases 9. In the present paper we report 4 cases of SnGCT treated via pure endoscopic or endoscopic-assisted resection. A review of the literature on this topic was also carried out.
Materials and methods
The current study is a retrospective analysis of primary SnGCT in patients admitted and treated with curative intent at the Unit of Otorhinolaryngology, ASST Grande Ospedale Metropolitano Niguarda, Milan, Italy, and the Unit of Otorhinolaryngology, ASST Spedali Civili di Brescia, Brescia, Italy, from 2001 to 2024. The work up of the patients consisted in radiological imaging with cone beam computed tomography (CBCT) and contrasted enhanced magnetic resonance imaging (MRI). Definitive pathologic diagnosis was established by consistent histology and immunohistochemistry. The information was extrapolated to report epidemiologic and clinal data, surgical approach and follow-up.
A comprehensive clinical review of the literature was also carried out on Scopus, PubMed/MEDLINE, Cochrane Library, Embase, ResearchGate and Google Scholar databases using relevant keywords, phrases, and MeSH terms. An example of a search strategy was the one used for PubMed/MEDLINE: “giant cell tumour” [All Fields] AND (nasal sinuses [MeSH Term] OR nasal fossa [MeSH Terms] OR surgery, endoscopic surgery [MeSH Terms]). References were scrutinised for additional articles.
Results
In the series were included 4 cases of SnGCT surgically treated with curative intent in the 2 referral centres.
Case presentation
CASE 1
A 12-year-old girl presented with bilateral worsening nasal obstruction lasting a few months. The patient had a family history of cherubism. The endoscopic examination showed a submucosal regular lesion centred on the lower third of the nasal septum, with the root on the nasal floor. The patient was studied with CBCT followed by a contrast enhanced MRI. The biopsy was avoided because the lesion appeared hypervascularised. An expert radiologist hypothesised the benign nature of the lesion, and the possible consistence with SnGCT, also considering the family history of the patient. A complete resection was obtained via pure endoscopic transnasal surgery. The definitive histological diagnosis was consistent with SnGCT. The histopathological analysis with routine haematoxylin and eosin staining revealed spindle-shaped elements with numerous multinucleated osteoclast-like giant cells, with haemosiderin pigment and haemorrhage, incorporating bone tissue. Immunohistochemical studies were performed with appropriate positive and negative controls. The giant cells were strongly positive for macrophage marker CD68. There was no major bleeding or postoperative haemorrhagic complications. At 6-year follow-up the patient resulted free of disease. Radiology and histology of the patient are reported in Figure 1.
CASE 2
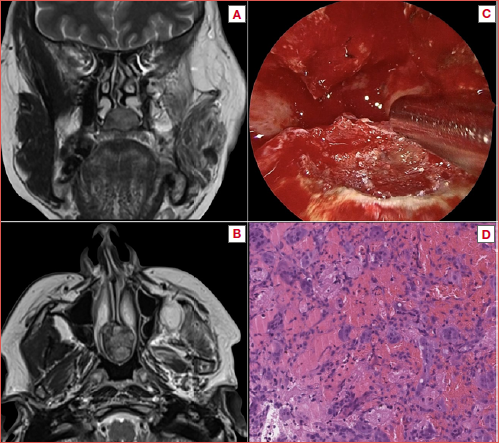
A 45-year-old woman in follow-up with a previous history of a surgically treated GCT of the mandible, had a radiological finding of nasal mass at the contrast-enhanced MRI. She therefore completed the work up with a CBCT and an endoscopic examination: in consideration of the previous medical history, the tumour was classified as a SnGCT of the nasal rostrum with a large root on the nasal floor. The periphery of the lesion appeared hyperostotic, and thus a preoperative biopsy was avoided. The resection of the tumour was completed with a pure endoscopic transnasal approach, with posterior septectomy, and drilling of the basement of the tumour on the nasal floor. The definitive histologic diagnosis was consistent with GCT. The histopathological analysis with routine haematoxylin and eosin stains revealed non-capsulated proliferation of spindle-shaped elements with numerous multinucleated osteoclast-like giant cells, with haemosiderin pigment and haemorrhage, incorporating bone tissue. Immunohistochemical studies were performed with appropriate positive and negative controls. The giant cells were strongly positive for macrophage marker CD68. The patient is free of disease after 4 years of follow-up. Radiological, clinical and histological images of the patient are shown in the Cover figure.
CASE 3
An 18-year-old male patient presented with bilateral nasal obstruction persisting for 2 months. He had undergone surgery for nasal polyposis elsewhere 2 years earlier; histopathological report was not retrieved. Contrast-enhanced MRI revealed a 4 cm lesion originating from the posterior nasal septum, obstructing the nasal cavities. A biopsy was consistent with SnGCT, and an endoscopic transnasal resection with septectomy was carried out. The final diagnosis confirmed a SnGCT of the nasal rostrum. Histopathology showed spindle-shaped elements with numerous multinucleated osteoclast-like giant cells, haemosiderin pigment, haemorrhage, and bone tissue incorporation. Immunohistochemistry revealed PGM-1 and CD1673 positivity in giant cells, partial smooth muscle actin positivity in spindle cells, and negative S100 protein. Postoperative course was uneventful. At the 2-year follow-up, the patient remained disease-free.
CASE 4
A 70-year-old woman with Paget’s disease was referred for recurrent SnGCT. Primary disease was located in the right maxillary sinus; during follow up, MRI diagnosed a mass in the right nasal fossa suspicious for disease relapse. She underwent centripetal subperiosteal endoscopic resection of the right nasal fossa, including medial maxillectomy, maxillary mucosa and bony roof, ethmoidectomy (including lamina papiracea), lacrimal pathway, and pterygopalatine fossa content. Histology confirmed the diagnosis of SnGCT with numerous multinucleated osteoclastic giant cells and mononucleated fibro-histiocytic cells, along with sclerosis, haemosiderin deposits, and xanthomatous histiocytes. Adjacent bone tissue alterations were consistent with Paget’s disease.
During follow-up, several recurrences occurred. At 2 months, a lesion consistent with SnGCT was removed at the occipital bone. Then, 2 years later the disease relapsed at both right maxillary and occipital sites. The maxillary recurrence was managed with an endoscopic approach requiring resection of the periorbita and the ethmoid roof. One year later, SnGCT recurred in the left nasal fossa requiring endoscopic centripetal resection with ethmoidectomy and medial maxillectomy.
In view of the concomitant Paget disease (a risk factor for SnGCT), it was impossible to assess whether the localisations at occipital site and left nasal fossa were metastatic disease or second primaries. The last MRI (2 years after the last procedure) showed a 17 mm solid mass in the left nasal fossa extending from the orbital floor to the nasal cavity roof, displacing the superior turbinate with possible intraorbital extension. A 4 cm mass infiltrated the left retroauricular and nuchal soft tissues, destroying the outer diploe at the temporo-occipital junction. The hard palate lesion extended beyond the midline, lifting the nasal cavity floor mucosa and involving the lower nasal septum. The patient refused further surgical treatment and was lost at follow-up.
The patients’ data are summarised in Table I.
The patient and the legal guardians of Case 1 agreed to the publication of the clinical data.
Literature search and results
A total of 25 studies with 32 patients were included in this review (Tab. II). The male to female ratio was 0.37. Ten (31%) patients presented SnGCT with multiple locations, 9 (28%) presented a sphenoid sinus involvement, 4 (12.5%) maxillary sinus, 5 (15.6%) nasal cavities, 3 (9.3%) frontal sinus, and 1 (3%) ethmoid. Most patients (53%) underwent exclusive surgical resection, although only 4 (12.5%) were treated with pure endoscopic surgery, while 47% of cases integrated surgery with adjuvant denosumab or radiation therapy. Local recurrence was documented in 12.5% of cases, while distant metastasis in 3%. In particular, 79% of pure surgical excisions obtained free margins of resection (R0) and none had local recurrence. On the other hand, the patients who underwent multimodal treatment had positive margins (R1 or R2) in 70% of cases and 33% of these had local recurrence.
Discussion
Differential diagnosis of sinonasal benign and indolent tumours continues to be demanding in consideration of the large variety of histologies. However, it is well known that benign masses are commonly included in 3 main groups: hypervascularised malformations as the juvenile angiofibroma, fibro-osseous lesions, and sinonasal papillomas. To these, several types of mesenchymal locally aggressive tumours, such as desmoid type fibromatosis, solitary fibrous tumour, and sinonasal glomangioperycitoma must be added. When a tumour falls outside these groups, the diagnosis can be very challenging, and integration between familiar and medical history, and radiology becomes essential. GCT is a benign primary tumour of the bone, extremely rare in the sinonasal district, accounting for less than 2% of all GCTs, the majority reported in multiple sites. However, it has some peculiarities that make it of interest in the differential diagnosis of sinonasal masses. Indeed, SnGCT can be misdiagnosed as fibro-osseous lesions, hypervascularised neoplasms, or other borderline mesenchymal tumours. Most of the available literature about this entity in the sinonasal region consists of case reports. During our search, only 32 reported cases of SnGCT were found (Tab. II). Because of the very indolent symptoms, these tumours can be diagnosed at an advanced stage, which can affect the choice of the treatment since surgery can have a non-deniable rate of complications and cosmetic impact.
Clinical and radiological work up
Symptoms like nasal obstruction, frequent nasal infection and epistaxis are associated with multiple conditions of nasal and paranasal sinuses. Radiologic findings in SnGCT are characterised by varying degrees of bone destruction and these lesions tend to be locally aggressive 10. CBCT/CT and MRI are considered the ideal tools to characterise and diagnose SnGCT. On CBCT/CT, this type of tumour often appears as a soft tissue density mass with destructive expansion into the bone, without cortex involvement 11. MRI provides better characterisation of the lesion through its higher contrast resolution and multiple sequences 11. SnGCT usually demonstrates low to intermediate signal intensity on T1 sequencing, hypointensity on T2 and enhance with gadolinium 12.
The diagnosis is most frequently obtained postoperatively because a pretreatment biopsy may be difficult in view of hypervascularity, site, or ossification of the lesion 13. Nevertheless, if the lesion is easily accessible, it is often preferable to perform a preoperative biopsy (Case 3).
Histologic features
The histological appearance of SnGCT consists in numerous large multinucleated giant cells dispersed in a background stroma of mononuclear spindle cells and monocytes. The monocyte nuclei have features identical to those of the larger osteoclast-like giant cells, which can contain large number of these nuclei (> 50). However, the histological pattern can vary and can contain areas of cystic degeneration, haemorrhage, haemosiderin deposition, occasional mitotic figures, or increased spindle cell stroma. Various conditions show multinucleated osteoclast-like giant cells in a background of oval to spindle-shaped mononuclear cells. These include giant cell granulomas (GCGs), brown tumour of hyperparathyroidism and cherubism. The differential diagnosis between GCTs and GCGs is usually guided by symptomatology (reported in 72% of GCTs while only 15% of GCGs) and the distribution pattern of giant cells in the stroma (homogeneously dispersed in 80% of GCTs versus grouped in clusters in 86.7% of GCGs). The most important difference between these lesions is the prognosis, GCTs being more aggressive and with a higher risk of recurrence 6,12. Moreover, the gene mutated in cherubism has been mapped to chromosome 4p16.3 14. Missense mutations are observed in the SH3BP2 gene mainly clustered in exon 9 15,16. Recently, a somatic mutation of this gene was described in a case of GCT 17.
Treatment
An endoscopic endonasal approach should be preferred when feasible. In fact, for both benign and malignant sinonasal tumours, the endoscopic approach is considered the gold standard of treatment, as it allows for radical resection even in cases of locally advanced disease and reduces the complications associated with open approaches. In selected cases, the resection must be integrated with an open approach 18. The resection of SnGCT can be compared to that of other fibro-osseous lesions of the sinonasal region. The pathological tissue needs to be excised taking into consideration the morbidity of the procedure. In some extended cases involving noble structures a planned R2 excision could be necessary. In cases of complete tumour removal, adjuvant radiation therapy is not indicated 19. On the other hand, in case of involvement of vital structures, a planned R2 resection can be unavoidable. If total tumour excision is not obtainable, subtotal excision plus radiotherapy results in acceptable tumour control rates. For many years pharmacological therapy was not considered in the treatment of SnGCT 19. However, knowing that giant cells in SnGCT overexpress the receptor activator of nuclear factor kappa-β ligand (RANKL) 20,21 has led to the clinical application of denosumab, a human monoclonal antibody that reduces osteolysis, inhibiting RANKL. This treatment is mostly utilised in patients with unresectable or metastatic disease, as it reduces tumour burden, improves symptoms, and helps prevent tumour recurrence. A series of clinal trials and case reports have shown that denosumab can control the disease and reduce the extension of surgery in patients with GCT 22. In a neoadjuvant setting, it may promote en bloc resection because of the development of a calcified rim around the tumour and its soft tissue component. However, in these studies the median follow-up was roughly one year, which is too short to demonstrate efficacy and safety in complicated cases. Recent studies reported a higher local recurrence rate in patients treated with neoadjuvant denosumab, possibly because of more limited surgical resections 23-25.
Conclusions
SnGCT is a benign but locally active or aggressive bone neoplasm that typically presents as a lytic lesion. The clinical behaviour of GCT is unpredictable, and it tends to recur locally and even metastasise. In the nose and paranasal sinuses it is an exceedingly rare entity, but it should be considered in the differential diagnosis with a large variety of histologies, such as fibro-osseous lesions, hypervascularised masses, and locally invasive mesenchymal tumours. Surgery is treatment of choice for resectable lesions, with endoscopic transnasal surgery being first-line whenever feasible. The use of the monoclonal antibody denosumab is an option in selected cases (large masses, recurrence non amenable to surgery, unfit patients for surgery); however, larger trials are needed to evaluate the risks and benefits of this treatment.
Conflict of interest statement
The authors declare no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author contributions
AC, RA: study concepts; RA, DM: study design; AC, FDP, CLM: data acquisition; AB, RA, AC, DM: quality control of data and algorithm; AC, CLM, DM: data analysis and interpretation; AC, RA, DM: manuscript preparation; DM, RA, AB, HI: manuscript editing; CP, AGD, LC, DM, RA: manuscript review.
Ethical consideration
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. All authors have read and approved the manuscript.
History
Received: April 21, 2025
Accepted: October 17, 2025
Figures and tables
Figure 1. Case 1 from the series. A) CBCT coronal view showing a soft tissue density expansive mass with destructive progression into the palate; B) Contrast-enhanced MRI T2-weighted coronal sequence showing the hypervascularity of the lesion; C) Haematoxylin and eosin stains showing spindle-shaped elements with numerous multinucleated osteoclast-like giant cells, with haemosiderin pigment and haemorrhage, incorporating bone tissue; D) Immunohistochemistry: the giant cells were strongly positive for macrophage marker CD68.
| Patient | Age (years) | Gender | Site of tumor | Type of surgery | State | Follow-up (months) |
|---|---|---|---|---|---|---|
| 1# | 12 | Female | Nasal cavity | Pure endoscopic | NED | 74 |
| 2# | 45 | Female | Nasal cavity | Pure endoscopic | NED | 48 |
| 3# | 18 | Male | Nasal cavity | Pure endoscopic | NED | 24 |
| 4# | 70 | Female | Multiple | Endoscopic-assisted | AWD | 96 (LAF) |
| NED: no evidence of disease; AWD: alive with disease; LAF: lost at follow-up. | ||||||
| Study | Year | Study design | Patients | Gender | Site | Treatment | Margins | Reoccurrence | Metastasis |
|---|---|---|---|---|---|---|---|---|---|
| Peimer26 | 1954 | Case series | 1# | M | Maxillary sinus | Open surgery | R0 | No | No |
| 2# | M | Multiple sites | Open + adjuvant therapy | R2 | No | No | |||
| Burnam 27 | 1970 | Case report | 1# | F | Multiple sites | Open surgery | R0 | No | No |
| Sgouras 28 | 1971 | Case report | 1# | M | Nasal cavity | Open surgery | NS | No | No |
| Fu10 | 1974 | Case series | 1# | F | Maxillary sinus | Open surgery | R0 | No | No |
| 2# | F | Maxillary sinus | Open surgery | R0 | No | No | |||
| 3# | F | Frontal sinus | Open surgery | R0 | No | No | |||
| 4# | M | Multiple sites | Open surgery | R2 | Yes | No | |||
| Hlaváček 29 | 1974 | Case series | 1# | F | Frontal sinus | Open surgery | R0 | No | No |
| 2# | M | Multiple sites | Open surgery | R0 | No | No | |||
| Handler 30 | 1982 | Case report | 1# | F | Multiple sites | Open surgery | R0 | No | No |
| Wolfe 31 | 1983 | Case series | 1# | F | Sphenoid sinus | Pure endoscopic + adjuvant therapy | R0 | No | No |
| 2# | F | Multiple sites | Open + adjuvant therapy | R0 | No | No | |||
| 3# | M | Sphenoid sinus | Pure endoscopic + adjuvant therapy | R0 | No | No | |||
| Anand 32 | 2001 | Case report | 1# | M | Nasal cavity | Open surgery | R0 | No | No |
| Chan 33 | 2003 | Case report | 1# | M | Multiple sites | Pure endoscopic | R0 | No | No |
| McKinney 34 | 2005 | Case report | 1# | F | Sphenoid sinus | Open surgery | R2 | Yes | Yes |
| Tuluc 35 | 2006 | Case report | 1# | F | Nasal cavity | Pure endoscopic | R0 | No | No |
| Esposito 36 | 2006 | Case report | 1# | F | Sphenoid sinus | Pure endoscopic + adjuvant therapy | R2 | No | No |
| Company 37 | 2009 | Case report | 1# | M | Sphenoid sinus | Open surgery | R2 | Yes | No |
| Battoo 38 | 2010 | Case report | 1# | F | Sphenoid sinus | Open + adjuvant therapy | R1 | No | No |
| Huang 39 | 2013 | Case report | 1# | F | Sphenoid sinus | Pure endoscopic | R0 | No | No |
| Goto 40 | 2016 | Case report | 1# | M | Sphenoid sinus | Pure endoscopic + adjuvant therapy | R2 | No | No |
| Rodriguez 41 | 2017 | Case report | 1# | F | Frontal sinus | Open surgery | R0 | No | No |
| Tonari 42 | 2017 | Case report | 1# | F | Multiple sites | Pure endoscopic + adjuvant therapy | R2 | Yes | No |
| Yip 43 | 2018 | Case report | 1# | F | Ethmoid sinus | Open + adjuvant therapy | R1 | No | No |
| Rosa 44 | 2018 | Case report | 1# | M | Maxillary sinus | Open + adjuvant therapy | R2 | Yes | No |
| Pousti 45 | 2020 | Case report | 1# | F | Multiple sites | Pure endoscopic + adjuvant therapy | R2 | No | No |
| Aassouani 46 | 2022 | Case report | 1# | F | Multiple sites | Pure endoscopic + adjuvant therapy | NS | No | No |
| Hu 47 | 2022 | Case report | 1# | M | Sphenoid sinus | Pure endoscopic | R2 | No | No |
| Serafini 48 | 2023 | Case report | 1# | F | Nasal cavity | Pure endoscopic | R0 | No | No |
| Linxweiler 49 | 2024 | Case report | 1# | F | Nasal cavity | Pure endoscopic | R0 | No | No |
| M: male, F: female, NS: non specified. | |||||||||
References
- Campanacci M, Baldini N, Boriani S. Giant-cell tumor of bone. 1987;69:106-114.
- Larsson S, Lorentzon R, Boquist S. Giant-cell tumor of bone. A demographic, clinical, and histopathological study of all cases recorded in the Swedish Cancer Registry fot the years 1958 through 1968. J Bone Joint Surg Am. 1975;57:167-173.
- Baena-Ocampo Ldel C, Ramirez-Perez E, Linares-Gonzalez L. Epidemiology of bone tumors in Mexico City: retrospective clinicopathologic study of 566 patients at a referral institution. Ann Diagn Pathol. 2009;57:167-173. doi:https://doi.org/10.1016/j.anndiagpath.2008.07.005
- Werner M. Giant cell tumor of bone: morphological, biological and histogenetical aspects. Int Orthop. 2006;30:484-489. doi:https://doi.org/10.1007/s00264-006-0215-7
- Park S, Chung S, Lim J. Giant cell tumor of the mandible. Clin Exp Otorhinolaryngol. 2012;5:49-52. doi:https://doi.org/10.3342/ceo.2012.5.1.49
- Johnston J. Muscoloskeletal tumors OKU: AAOS. Giant cell tumor of bone. Published online 2002:113-118.
- Campanacci M, Giunti A, Olmi R. Metaphyseal and diaphyseal localization of giant cell tumors. Chir Organi Mov. 1975;62:29-34.
- Pai S, Lalitha R, Prasad K. Giant cell tumors of the temporal bone – A case report. BMC Ear Nose Throat Disord. 2005;5. doi:https://doi.org/10.1186/1472-6815-5-8
- Cheng J, Johnston J. Giant cell tumor of bone. Prognosis and treatment of pulmonary metastases. Clin Orthop Relat Res. 1997;338:205-214. doi:https://doi.org/10.1097/00003086-199705000-00027
- Fu Y-S, Perzin K. Non-epithelial tumors of the nasal cavity, paranasal sinuses, and nasopharynx: a clinicopathologic study. II. Osseous and fibro-osseous lesions, including osteoma, fibrous dysplasia, ossifying fibroma, osteoblastoma, giant cell tumor, and osteosarcoma. Cancer. 1974;33:1289-1305. doi:https://doi.org/10.1002/1097-0142(197405)33:5<1289::AID-CNCR2820330514>3.0CO;2-P
- Lee H, Lum C. Giant-cell tumor of the skull base. Neuroradiology. 1999;41:305-307. doi:https://doi.org/10.1007/s002340050753
- Murphey M, Nomikos G, Flemming D. Imaging of giant cell tumor and giant cell granuloma of bone: radiologic-pathologic correlation. Radiographics. 2001;21:1283-1309. doi:https://doi.org/10.1148/radiographics.21.5.g01se251283
- Commins D, O’Malley S, Athansou N. Giant cell tumor of the hyoid-first reported case. J Laryngol Otol. 1999;113:566-568. doi:https://doi.org/10.1017/s0022215100144500
- Bell S, Shaw M, Jou Y. Identification and characterization of the human homologue of SH3BP2, an SH3 binding domain protein within a common region of deletion at 4p16.3 involved in bladder cancer. Genomics. 1997;44:163-170. doi:https://doi.org/10.1006/geno.1997.4849
- Ueki Y, Tiziani V, Santanna C. Mutations in the gene encoding c-Abl-binding protein SH3BP2 cause cherubism. Nat Genet. 2001;28:125-126. doi:https://doi.org/10.1038/88832
- De Lange J, van Maarle M, van den Akker H. A new mutation in the SH3BP2 gene showing reduced penetrance in a family affected with cherubism. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:378-381. doi:https://doi.org/10.1016/j.tripleo.2006.05.012
- Carvalho V, Perdigão P, Amaral F. Novel mutations in the SH3BP2 gene associated with sporadic central giant cell lesions and cherubism. Oral Diseases. 2009;15:106-110. doi:https://doi.org/10.1111/j.1601-0825.2008.01499.x
- Balke M. Giant cell tumor of the axial scheleton. Sarcoma. 2012;2012. doi:https://doi.org/10.1155/2012/410973
- Freeman J, Oushy S, Schowinsky J. Invasive giant cell tumor of the lateral skull base: a systematic review, meta-analysis, and case illustration. World Neurosurg. 2016;96:47-57. doi:https://doi.org/10.1016/j.wneu.2016.05.086
- Branstetter D, Manivel J. Denosumab induces tumor reduction and bone formation in patients with giant-cell tumor of bone. Clin Cancer Res. 2012;18:4415-4424. doi:https://doi.org/10.1158/1078-0432.CCR-12-0578
- Huang L, Xu J, Wood D. Gene expression of osteoprotegerin ligand, osteoprotegerin, and receptor activator of NF-kappaB in giant cell tumor of bone: possible involvement in tumor cellinduced osteoclast-like cell formation. Am J Pathol. 2000;156:761-767. doi:https://doi.org/10.1016/s0002-9440(10)64942-5
- van der Heijden L. Giant cell tumor of bone in the denosumab era. Eur J Cancer. 2017;77:75-83. doi:https://doi.org/10.1016/j.ejca.2017.02.021
- Errani C, Tsukamoto S, Leone G. Denosumab may increase the risk of local recurrence in patients with giantcell tumor of bone treated with curettage. J Bone Joint Surg Am. 2018;100:496-504. doi:https://doi.org/10.2106/JBJS.17.00057
- Traub F, Singh J, Dickson B. Efficacy of denosumab in joint preservation for patients with giant cell tumour of the bone. Eur J Cancer. 2016;59:1-12. doi:https://doi.org/10.1016/j.ejca.2016.01.006
- Agarwal M, Gundavda M, Gupta R. Does denosumab change the giant cell tumor treatment strategy? Lessons learned from early experience. Clin Orthop Relat Res. 2018;476:1773-1782. doi:https://doi.org/10.1007/s11999.0000000000000243
- Peimer R. Benign giant-cell tumors of skull and nasal sinuses. Arch Otolaryngol Head Neck Surg. 1954;60:186-193. doi:https://doi.org/10.1001/archotol.1954.00720010192008
- Burnam A, Benson J, Cohen I. Giant cell tumor of ethmoid sinuses: diagnostic dilemma. Laryngoscope. 1970;80:1415-1424. doi:https://doi.org/10.1002/lary.5540890906
- Sgouras N, Georgiades D. Giant cell tumor of nasal bones. Plast Reconstr Surg. 1971;48:184-186. doi:https://doi.org/10.1097/00006534-197108000-00021
- Hlaváček V, Jolma V. Giant cell tumors of bone in the E.N.T. organs: report of two cases in the frontal sinus. Acta Otolaryngol. 1974;77:374-380. doi:https://doi.org/10.3109/00016487409124639
- Handler S, Savino P, Peyster R. Giant cell tumor of the ethmoid sinus: an unusual cause of proptosis in a child. Otolaryngol Head Neck Surg. 1982;90:513-515. doi:https://doi.org/10.1177/019459988209000430
- Wolfe J, Scheithauer B, Dahlin D. Giant-cell tumor of the sphenoid bone. Review of 10 cases. J Neurosurg. 1983;59:322-327. doi:https://doi.org/10.3171/jns.1983.59.2.0322
- Anand T, Kumar D, Kumar S. Giant cell tumor of hard palate. Indian J Otolaryngol Head Neck Surg. 2001;53:299-300. doi:https://doi.org/10.1007/BF02991553
- Chan J, Gannon F, Thompson L. Malignant giant cell tumor of the sphenoid. Ann Diagn Pathol. 2003;7:100-105. doi:https://doi.org/10.1053/adpa.2003.50020
- McKinney A, Reichert P, Short J. Metachronous, multicentric giant cell tumor of the sphenoid bone with histologic, CT, MR imaging, and positron-emission tomography/CT correlation. AJNR Am J Neuroradiol. 2005;27:2199-2201.
- Tuluc M, Zhang X, Inniss S. Giant cell tumor of the nasal cavity: case report. Eur Arch Otorhinolaryngol. 2006;264:205-208. doi:https://doi.org/10.1007/s00405-006-0143-6
- Esposito F, Kelly D, Vinters H. Primary sphenoid sinus neoplasms: a report of four cases with common clinical presentation treated with transsphenoidal surgery and adjuvant therapies. J Neurooncol. 2006;76:299-306. doi:https://doi.org/10.1007/s11060-005-7285-z
- Company M. Giant cell tumor of the sphenoid. Arch Neurol. 2009;66. doi:https://doi.org/10.1001/archneurol.2008.509
- Battoo A, Salih S, Unnikrishnan A. Oncogenic osteomalacia from nasal cavity giant cell tumor. Head Neck. 2012;34:454-457. doi:https://doi.org/10.1002/hed.21562
- Huang P, Lee C, Chang P. Giant cell tumor of the sphenoid bone occurring during pregnancy: successful tumor extirpation via endoscopic transnasal transsphenoidal surgery. Clin Neurol Neurosurg. 2013;115:222-226. doi:https://doi.org/10.1016/j.clineuro.2012.04.02
- Goto Y, Furuno Y, Kawabe T. Treatment of a skull-base giant cell tumor with endoscopic endonasal resection and denosumab: case report. J Neurosurg. 2017;126:431-434. doi:https://doi.org/10.3171/2016.3.JNS152802
- Rodriguez Cunha B, Fontalvo Martin M, Canavezi Indiani J. Giant cell tumor of the frontal sinus: a typical finding in an unlikely location. Radiol Bras. 2017;50:414-415. doi:https://doi.org/10.1590/0100-3984.2016.0060
- Tonari M, Matsuo J, Oku H. A case of childhood-onset giant cell tumor that caused optic nerve atrophy in both eyes. Case Rep Ophthalmol. 2017;8:301-307. doi:https://doi.org/10.1159/000477199
- Yip C-M, Lee H-P, Hsu S-S. Left orbital roof giant cell tumor of bone: a case report. Surg Neurol Int. 2018;9. doi:https://doi.org/10.4103/sni.sni_467_17
- Rosa D, Dias R, Salvador J. Recurrent giant cell tumour of the maxillary sinus and pterygoid process treated with denosumab. BMJ Case Rep. Published online 2018. doi:https://doi.org/10.1136/bcr-2018-225095
- Pousti B, Andera L, Haugen T. Sphenoid sinus giant cell tumor: a case report and literature review. ORL. 2020;82:168-174. doi:https://doi.org/10.1159/000506207
- Aassouani F, El Bouardi N, Charifi Y. A rare case of sphenoid giant cell tumor: case report & review of imaging features post short-term denosumab treatment. Radiol Case Rep. 2022;17:3830-3834. doi:https://doi.org/10.1016/j.radcr.2022.07.040
- Hu S, Cheng S, Wu Y. A large cavernous sinus giant cell tumor invading clivus and sphenoid sinus masquerading as meningioma: a case report and literature review. Front Surg. 2022;9. doi:https://doi.org/10.3389/fsurg.2022.861739
- Serafini E, Basso M, Melchiorri C. Multifocal giant cell tumor of the nasal fossa: case report and literature review. Laryngoscope. 2024;134:2774-2778. doi:https://doi.org/10.1002/lary.31220
- Linxweiler M, Remke M, Straub B. Giant cell tumor of the inferior turbinate in a 12-year-old child: first case report. OTO Open. 2024;8. doi:https://doi.org/10.1002/oto2.165
Downloads
License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright
Copyright (c) 2026 Società Italiana di Otorinolaringoiatria e chirurgia cervico facciale
How to Cite
- Abstract viewed - 350 times
- PDF downloaded - 83 times



