Reviews
Vol. 46: Issue 1 - February 2026
Haemangiopericytoma and solitary fibrous tumour of the parotid gland: a scoping review
Summary
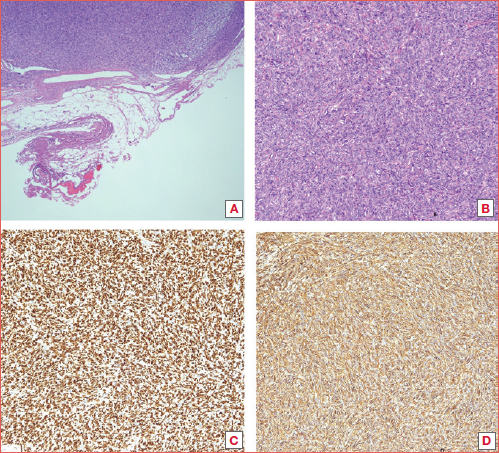
Salivary gland tumours represent about 3-10% of head and neck tumours, with the parotid gland being the most affected. The World Health Organization classification of these tumours is continually evolving due to their diverse histology and biological behaviour. Haemangiopericytoma (HPC) and solitary fibrous tumour (SFT) have been reclassified as a single entity characterised by NAB2-STAT6 gene rearrangement, typically presenting as slow-growing, painless masses in the parotid region, and immunohistochemistry is essential for diagnosis. This study aims to examine the clinical characteristics of these tumours to better understand their biological behaviour. Following PRISMA 2020 guidelines, we reviewed the literature using the search string (haemangiopericytoma), OR (solitary fibrous tumor) AND (parotid gland). Of the initially collected 201 papers, 49 were selected for detailed analysis, focusing on data from 53 patients, all of whom underwent surgery. Of these patients, 11.3% (n = 6) were diagnosed with malignancy, while 3 more were suspected to have malignancy. Our findings suggest that SFT morphology predominates among lesions, but malignancy is more often associated with features of HPC. Further comprehensive study is needed to gather additional data on their biological behaviour.
Introduction
Salivary gland tumours represent approximately 3-10 % of head and neck tumours, wherein the parotid gland is the most affected, and around 25% of such tumours are malignant 1. Because of their heterogeneous histology and biological behaviour, the World Health Organization (WHO) classification of these tumours continues to change. In this context, haemangiopericytoma (HPC) and solitary fibrous tumour (SFT) are rare entities. Despite historically being considered two different entities, the 4th Edition of the WHO classifies them as fibroblastic tumours characterised by haphazardly arranged spindled-to-ovoid cells, with prominent staghorn vasculature and NAB2-STAT6 gene rearrangement 2.
HPC was described for the first time in 1942 3; it originates from the pericytes of Zimmerman surrounding the capillaries and postcapillary venules 4, and can be found in the head and neck region in 15-25% of cases 5.
SFTs are composed of pleomorphic spindle cells, are distributed and patternless, and are situated within variably dense collagenous stroma. The most common sites for these tumours in the head and neck are the sino-nasal tract, orbit, oral cavity, and salivary glands 6.
Tumours with high mitotic count, hypercellularity, necrosis, and atypia (round cell and anaplastic morphology, or nuclear pleomorphism) are considered malignant 7. The NAB2-STAT6 fusion gene is a unique molecular feature of the HPC/SFT family, present in up to 100% of cases 8.
Immunohistochemistry is very important for correct diagnosis. The markers used include CD34, Bcl2, CD99, and vimentin expression in the absence of actin, desmin, S100 protein, and epithelial cells 9. Immunostaining for STAT6 rearrangement is also available in pathology laboratories 10,11.
These tumours typically present as slow-growing, painless masses in the parotid region. Due to their slow growth, symptoms are not always noticeable in the early stages. While they are generally considered benign, there have been instances of malignant behaviour, though there is currently limited knowledge about this type of tumour due to its rarity.
On CT scans, they are both often homogeneous in appearance, although cystic areas, calcifications, myxoid degeneration, or haemorrhage may be present, particularly in large tumours 12. They tend to displace rather than invade surrounding tissues, and enhance after contrast administration. On MRI, HPC/SFT displays low T1 signal intensity and variable T2 signal 13, and enhances intensely after intravenous gadolinium administration 14 despite these characteristics are not pathognomonic for HPC/SFT. While HPC/SFT demonstrate benign behaviour, follow-up is essential to detect any sign of local recurrence early on. A tendency to both local and distant recurrence has been described 15.
Treatment requires surgical removal, with approaches varying depending on location and extent of involvement 16. This study endeavours to explore the clinical characteristics of these tumours to better understand their biologic potential and their transformation into a malignant entity through a review of the literature (Cover figure).
Materials and methods
This review was conducted with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 guidelines (Fig. 1)17. The research was carried out using the PubMed and Scopus database with the following research string: [(haemangiopericytoma) OR (solitary fibrous tumor)] AND (parotid gland). Two reviewers (C.L. and E.S.) performed the literature search and the abstract and full text reading. All the articles found were included, without any period of restriction. The last research on the database was performed in April 2024.
The inclusion criteria for first abstract reading selection were:
- abstracts in English;
- articles about haemangiopericytoma or solitary fibrous tumour;
- studies on diseases of the parotid gland.
The exclusion criteria for this phase were:
- abstract in other languages;
- articles treating different topics;
- diseases of other salivary glands.
After the first abstract based selection, a full text reading was performed, using additional criteria.
The inclusion criteria for the second selection phase were:
- case report or case series as study design;
- full text available;
- full text in English;
- studies on diseases of the parotid gland.
The exclusion criteria were:
- review;
- the full text not being available;
- the full text being in another language;
- studies on disease of the other salivary glands.
Two-hundred and one papers were selected. In total, 58 duplicated articles were eliminated, resulting in 143 articles being eligible for further analysis. First, an abstract reading selection was made according to the inclusion criteria, in which 68 abstracts were selected. After this, a further exclusion following full-text reading and the pre-established criteria was made, with 49 articles being selected (Fig. 1).
The selected articles were then analysed in detail, extracting data on study design; publication year; age and gender of the patients; imaging and histologic reports of the first parotid disease and its metastasis; the type of treatment and outcomes; potential recurrence and related treatment and follow-up status.
Results
Ten of the 49 articles selected (20%) were published before the year 2000. A total of 53 patients (28 males and 25 females) were reported in the selected papers, and the mean age at diagnosis was 49.3 years (range, 11-82), while four (7.6%) cases were paediatric. In total, 22 patients developed the disease in the right parotid gland, while for 30 patients it developed in the left (for one patient, data were missing). Clinical data of the sample are reported in Table I. All the selected articles are listed in Table II.
Regarding clinical presentation, 48 patients showed swelling in the parotid area, 5 complained of loco-regional pain, and in 2 of 5 cases the mass was situated in the deep lobe 21,25,55,56. Only one patient showed facial nerve impairment at the first medical examination; this patient was a 72-year-old woman with a 5-year history of a slowly enlarging left parotid mass and a diagnosis of malignant solitary fibrous tumour 56. In the paediatric cases, 3 of 4 patients were females, and swelling in the parotid area was seen in each.
With regard to preoperative assessments, 39 (73.5%) patients in total were referred to imaging (25 underwent CT and 14 underwent MRI), while 17 (32%) underwent ultrasound only. From a cytopathology perspective, most of the cases did not report preoperative fine-needle aspiration cytology (FNAC) evaluation and cytologic description. In 5 studies an inconclusive result was indicated, even if repeated in one case.
Of all the subjects, the vast majority – 49 in total – underwent surgery. Specifically, 11 underwent enucleoresection, 14 underwent superficial parotidectomy, 20 had total parotidectomy, and 4 had subtotal parotidectomy. A transnasal/transoral surgical approach was taken in one patient 4. However, treatment data were not disclosed for 3 cases of HPC 25,56.
Only one patient received primary chemotherapy (CHT) and subsequent surgery 21. After failure of CHT, a stylohamular approach was taken and a suprahyoid neck dissection was performed.
The histological diagnosis was HPC (30.2%) for 16 patients, 4 of whom (25%) had malignancy, while SFT was diagnosed in 37 patients (69.8%), of which 2 (5%) had malignancy; overall, the total number of malignant cases was 6 (11.3%). In 2 cases, tumours with the histological characteristic of “aggressivity in nature” 4,18 were described, and in one, undifferentiated spindle cells were reported 28. These 3 subjects each underwent RT and were diagnosed with HPC. Including these, the overall malignancy rate was 16.9%, and 43% (7/16) in HPC cases. All relevant histological markers are detailed in Table II.
Concerning malignant cases, one patient was disease-free for nearly 5 years after surgery. She subsequently developed pulmonary and hepatic metastases, which were surgically removed 36. The second patient with malignancy 21 died due to distant metastases, with no specification regarding the location or time of onset. In the third case, the tumour showed two different behaviours under microscopy: a histologically benign area of haemangiopericytoma was present and separated by a clear-cut border from a second area, in which the structure was that of a histologically malignant haemangiopericytoma. The patient developed fatal metastasis 20.
The fourth concerns a patient with a diagnosis of malignant HPC who died with recurrence of the disease after surgery 19, although distant metastases were not mentioned. In the recent fifth case, a young adult with a malignant SFT underwent surgery and RT with no signs of recurrence or distant metastasis at 11 months 63. The final case 56 relates to a case of malignant SFT, in which distant metastases were not mentioned during follow-up. Four patients (7.5%) had postoperative RT 18,63,28, and one underwent both CHT and RT 4.
Six patients (11.3%) had loco-regional recurrence of disease, while only one was malignant. In total, 4 of 53 patients (7.5%) developed distant metastasis, while 3 of 6 malignant cases (50%) and one uncertain case with peculiar characteristics presented metastasis at follow-up 18. The most common site of metastasis was the lungs, although other sites included the pleura, pelvis, muscles, body of vertebrae, retroperitoneum, and liver, with details shown in Table III.
The mean follow-up of the patients was 24.1 months, although in 12 (22%) cases this information was not available.
Discussion
To the best of our knowledge, this article presents the first systematic review of HPC/SFT within the parotid gland. The epidemiological characteristics confirmed by this review show a median age of incidence of 49.3 years, ranging from 11 to 82 years, and a male-to-female ratio of 28 to 25. However, the tumour incidence in the juvenile population and in females was not as low as expected. Indeed, it has been reported that around 5% of salivary gland tumours occur in patients under 18 years 66 of age, although this review shows a higher percentage, namely 7.6% of paediatric cases.
Concerning clinical features, the vast majority of cases herein presented a slow-growing mass in the parotid region. Grossly, these tumours were mostly well circumscribed, with a fibrous pseudocapsule and a cut surface ranging from firm and white to tan and fleshy for cellular variants. Histological analysis usually revealed a patternless distribution of ovoid- and spindle-shaped cells within a variably dense collagenous stroma for SFT morphology, or a branching and dilated staghorn-like vascular network for HPC.
STAT6 and CD34 have proven to be very useful markers. Before the advent of immunohistochemistry, diagnosis relied solely on microscopic analyses without the use of molecular markers.
The precise definition of malignant HPC/SFT remains undetermined. However, certain characteristics such as a high mitotic index, substantial tumour size, increased cellularity, the presence of immature tumour cells, and the occurrence of haemorrhagic or necrotic areas are indicative of a malignant nature of the lesion 5. While tumour characteristics included hyperchromatism and bone destruction, the set of histological data obtained from this review showed that the primary characteristic used for diagnosing malignant tumour was an increased number of mitoses.
The case series results collected herein suggest that SFT and HPC have different biological behaviour. In fact, SFT morphology represented the majority of lesions, but malignancy was mainly related to the HPC pattern, especially when taking into account the 3 cases of highly suspected malignancy. Moreover, one such case was paediatric. In other studies, it has been reported that childhood parotid neoplasms differ from those in adults, being primarily malignant but often presenting at lower grades 67,68.
As expected, surgical approaches tailored to each patient have been the treatment of choice, from superficial parotidectomy to extension to the parapharyngeal space 27. The extent of surgery should be determined by the size and location of the pathology and the structures it affects. Recurrence may occur due to incomplete resection, tumour seeding, within the near tissue, or distant haematogenous spread 16.
We found the recurrence rate to be higher than expected; of 53 patients, recurrence was recorded in 11.3%, and treatment options included surgery, RT, or a combined approach. Local recurrence was observed in most cases in SFT, and the mean time of onset was 24.2 months.
Künzel et al. 49 reported a case of SFT located in the left parotid gland which was removed with no clear margins, carried out in consideration of the tumour’s proximity to the carotid artery; the tumour relapsed after one year. Another case of recurrence was described by Chintale et al. 4 in which a large parotid HPC invading the right maxillary and masticator space relapsed locally after excision and was treated using RT and CHT. The recurrence rate appears to be correlated with tumour dimension, and most importantly the feasibility of achieving clear surgical margins.
Distant metastases were described in 4 cases and were linked to a malignant HPC histology, with the most common site being the lungs. Metastases had a mean time of onset of 31.2 months and were approached with surgery in one case and CHT in another, while for the other patients the chosen treatment was not disclosed. Currently, there are no strict guidelines for adjuvant treatment such as RT. The decision for adjuvant treatment depends on individual patient factors, mainly histological results, and clinical behaviour.
Follow-up is essential to detect any signs of malignancy or local recurrence early on, according to a multidisciplinary approach involving surgeons, oncologists, radiation oncologists, and pathologists. Due to the peculiar characteristics of HPC/SFT a tailored, multidisciplinary follow-up would be recommended in all cases and the role of pathologists appears important. Routine follow-up scans are generally not performed after surgery for benign tumours unless there are specific indications such as tumour rupture during surgery, spillage of cells into the parotid bed, or inability to achieve clear surgical margins. In these circumstances, MRI with gadolinium is preferred.
The scarcity of large case series and multicentric studies on these pathological entities has resulted in diverse treatment protocols and varying approaches in the management of patients with these tumours. The absence of comprehensive data impedes the development of standardised guidelines for their management, similar to other rare lesions in the head and neck region 69. Indeed, advances in immunohistochemistry and molecular discoveries have significantly improved our understanding of tumour classification and subtyping. The reclassification of HPC/SFT based on these new findings has likely led to more accurate diagnosis and identification of these tumours. However, due to the retrospective nature of some studies and a lack of comprehensive data, it is difficult to determine the number of misdiagnosed cases before the reclassification was made. The true incidence of HPC/SFT is underestimated due to the absence of immunohistochemistry and the inability to use molecular markers, especially in historical cases.
Conclusions
HPC/SFT is a mesenchymal tumour that rarely affects the head and neck region. This entity demonstrated different biological behaviour, as SFT represented the majority of lesions but malignancy was mainly related to HPC, especially considering the cases in which malignancy was strongly suspected. Surgery is the treatment of choice. When achieving optimal complete surgical excision, the risk of recurrence for SFT/HPC is relatively low. Regular follow-up is recommended in all cases due to the unpredictable behaviour of these neoplasms, where benign and malignant areas can coexist. When malignant behaviour is demonstrated, the risk of metastasis is high. Further comprehensive studies, particularly concerning histological and immunohistochemical characterisation, are essential to gather additional data and obtain more reliable and consistent results.
Conflict of interest statement
The authors declare no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author contributions
DM, LS, AE: conceived and designed the analysis; ES,CL: collected the data; AV, SP, RN: contributed data or analysis tools; SP, AE VPF: performed the analysis; CL, VC, GA: wrote the paper; SP, AV, RN: revised the paper; LS, DM, AE, SP: completed the final revision and approval of the draft.
Ethical consideration
Not applicable.
History
Received: April 23, 2025
Accepted: July 1, 2025
Figures and tables
Figure 1. PRISMA 2020 flow diagram of the literature selection process.
| Clinical presentation | Swelling in parotid region n = 48 (90.6%) |
| Pain n = 5 (9.4%) | |
| Paraesthesia n = 1 (1.9%) | |
| Facial nerve impairment n = 1 (1.9%) | |
| Mean age | 49.3 y (11-82) |
| Gender | 28 M (52.8%) |
| 25 F (47.2%) | |
| Surgery | Enucleoresection n = 11 (20.7%) |
| Superficial parotidectomy n = 14 (26.4%) | |
| Total parotidectomy n = 20 (37.7%) | |
| Subtotal parotidectomy n = 4 (7.5%) | |
| Transnasal-transoral approach n = 1 (1.9%) | |
| No data available n = 3 (5.6%) |
| Authors | Year | Cases | Gender | Age | Treatment of parotid disease | Histology | Marker (+) | Marker (-) | Adjuvant treatment | Recurence | Metastasis | Follow-up time, months/status |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neal et al. 18 | 1973 | 1 | M | 21 | TP | HPC malignant? | NA | NA | RT | yes | 48/DOD | |
| Katz 19 | 1975 | 1 | F | 69 | ER | Malignant HPC | NA | NA | yes | 168/DOD | ||
| Massarelli et al. 20 | 1980 | 1 | M | 64 | TP | Malignant HPC | NA | NA | yes | 16/DOD | ||
| Katsantonis et al. 21 | 1989 | 1 | M | 22 | CT | Malignant HPC | NA | NA | yes | 18/DOD | ||
| Carrillo et al. 22 | 1992 | 3 | F | 79 | TP | HPC | NA | NA | 18/AND | |||
| F | 38 | SP | HPC | 18/AND | ||||||||
| M | 27 | SP | HPC | 5/AND | ||||||||
| Lee et al. 23 | 1992 | 1 | M | 14 | SP | HPC | NA | NA | 24/AND | |||
| Whittam et al. 24 | 1993 | 1 | F | 82 | ER | HPC | NA | S100, epithelial markers | 12/AND | |||
| Hanau et al. 25 | 1994 | 1 | F | 73 | NA | SFT | VIM, CD34 | CK, CD31 | NA | |||
| Ferreiro et al. 26 | 1996 | 2 | M | 46 | SP | SFT | VIM, CD34 | - | 1/AND | |||
| F | 78 | SP | SFT | 12/AND | ||||||||
| Sato et al. 27 | 1998 | 1 | M | 52 | TP | SFT | VIM, CD34 | Desmin, S100, SMA | 12/AND | |||
| Mortelé et al. 65 | 2000 | 1 | M | 26 | TP | HPC | NA | NA | NA | |||
| Sawh et al. 28 | 2000 | 1 | F | 65 | STP | Undifferentiated spindle-cell neoplasm HPC | VIM, CD34 | SMA, desmin, S100, CK | RT | 24/AND | ||
| Mohammed et al. 29 | 2001 | 1 | F | 42 | ER | SFT | VIM, CD34 | CAM 5.2, AE1/AE3, EMA, S100, GFAP, SMA | 3/AND | |||
| Kumagai et al. 30 | 2002 | 1 | F | 47 | TP | SFT | VIM, CD34, bcl-2 | SMA, S100, CK, collagen type IV, CD117 | 14/AND | |||
| Sikes et al. 31 | 2003 | 1 | M | 62 | TP | HPC | NA | NA | 72/AND | |||
| Thompson et al. 32 | 2004 | 1 | F | 11 | TP | SFT | CD34, bcl-2 | S100, desmin, EMA, SMA | 24/AND | |||
| Gerhard et al. 33 | 2004 | 1 | F | 34 | SP | SFT | VIM, CD34 | Actin, SMA, S100 | 24/AND | |||
| Wiriosuparto et al. 34 | 2005 | 1 | M | 42 | TP | SFT | CD34, bcl-2, CD99 | AE1/AE3, CAM5.2, S100, calponin, p63 | NA | NA | ||
| Sreetharan et al. 35 | 2005 | 1 | M | 44 | TP | SFT | NA | NA | NA | |||
| Balaji et al. 36 | 2008 | 1 | F | 56 | TP | Malignant HPC | VIM, actin, CD34 | S100, GFAP | yes | 60/AWD | ||
| Cho et al. 37 | 2008 | 1 | M | 58 | TP | SFT | CD34, bcl-2, CD99 | S100, SMA, CK | 67/AND | |||
| Manglik et al. 38 | 2008 | 1 | M | 79 | SP | SFT | CD34, bcl-2, VIM | SMA, S100 and EMA | 3/AND | |||
| Bauer et al. 39 | 2011 | 1 | M | 66 | SP | SFT | CD34, bcl-2, VIM, CD99 | CD68, S100, CK, EMA, CAM5.2, SMA, actin, desmin, MYOD1, myogenin, CD117, GFAP, CD31, p63, p53, CD56, NFP, ALK-1 | 9/AND | |||
| Nowlin et al. 40 | 2011 | 1 | F | 28 | SP | HPC | CD34, FLI-1, CD68 | Desmin, SMA, S100, CK | 18/AND | |||
| Iyengar et al. 41 | 2011 | 1 | M | 49 | TP | SFT | VIM, CD34, CD99 | Desmin, S100, CD117, CK | NA | NA | ||
| Cristofaro et al. 42 | 2012 | 1 | M | 38 | ER | SFT | CD34, CD99, bcl-2, and VIM | CK, S100, SMA | 60/AND | |||
| Chen et al. 43 | 2013 | 1 | M | 33 | TP | HPC | CD34, CD99, bcl-2,VIM | CK, EMA, HMB45, S100, CD117, p63 | 12/AND | |||
| Sousa et al. 44 | 2013 | 1 | M | 32 | SP | SFT | CD34, CD99 and bcl-2 | CAM5.2, SMA and S100 | 10/AND | |||
| Apuhan et al. 45 | 2013 | 1 | M | 51 | ER | SFT | CD34 and bcl-2, CD99 | S100, Melan-A | NA | |||
| Liu et al. 46 | 2014 | 1 | M | 55 | ER | SFT | CD34, VIM, CD99, bcl-2 | S100, CK, SMA | 10/AND | |||
| Chis et al. 47 | 2014 | 1 | F | 67 | TP | SFT | VIM, CD34, bcl-2 | S100, SMA, GFAP | NA/AND | |||
| Alonso-Rodriguez et al. 48 | 2014 | 1 | F | 73 | ER | SFT | CD34, CD99, bcl-2 | AE/1, AE/2, EMA, S100 | Yes | 9/AND | ||
| Künzel et al. 49 | 2015 | 1 | F | 46 | STP | SFT | CD34, CD99, VIM, bcl-2 | CK, S100 | Yes | 19/AND | ||
| Yu et al. 50 | 2015 | 1 | M | 53 | SP | SFT | CD34, CD99, and bcl-2 | Pan-CK, EMA, SMA, desmin, S100, CD31, p63, ALK-1 | 24/AND | |||
| Gervasio et al. 51 | 2017 | 1 | M | 54 | SP | SFT | CD34, CD99, bcl-2 | NA | 24/AND | |||
| Rais et al. 52 | 2017 | 1 | M | 42 | TP | SFT | CD34, STAT6 | CK, SMA, S100, CD31 | 11/AND | |||
| Ralli et al. 53 | 2018 | 1 | F | 14 | ER | SFT | VIM, CD34, bcl-2, and CD99 | S100, desmin, SMA, myogenin | NA/AND | |||
| Lim et al. 54 | 2019 | 1 | M | 51 | STP | SFT | CD34, bcl-2, CD99, STAT6 | CK (AE1-3), CK14, SMA, CD21, CD35 | 3/AND | |||
| Chintale et al. 4 | 2020 | 1 | F | 14 | Transnasal and transoral approach | HPC “Agressive in nature” | NA | NA | CHT-RT | Yes | NA/AND | |
| Romano et al. 55 | 2020 | 1 | M | 50 | ER | SFT | CD34, bcl-2, STAT-6, CD99 | Actin, desmin, CK CAM | NA | |||
| Velez Torres et al. 56 | 2021 | 2 | M | 57 | NA | SFT | CD34 | Desmin, keratin, EMA, and p63 | NA | |||
| F | 72 | NA | Malignant SFT | NA | ||||||||
| Vitale et al. 57 | 2021 | 1 | F | 66 | TP | SFT | CD34, VIM, EMA, bcl-2, CD99 | CK, S100, HMB45, desmin, actin, CD117, CD31 | NA | |||
| Li et al. 58 | 2021 | 1 | M | 40 | ER | SFT | VIM, CD34, STAT6 | CD31, desmin, S100 | Yes | NA | ||
| Abdulhaleem et al. 59 | 2022 | 1 | F | 48 | TP | SFT | CD34, CD99, bcl-2, STAT6 | CK AE1/AE3, EMA, S100, CD21, CD23, SMA, CD31 | 12/AND | |||
| Goker et al. 60 | 2022 | 1 | F | 80 | ER | SFT | STAT6, CD34, CD99, bcl-2 | SMA, desmin, CK AE1/AE3 and ALK1 | 7/AND | |||
| Boschetti et al. 61 | 2022 | 1 | M | 52 | SP | SFT | STAT6, bcl-2, CD99 | SMA, calponin, CD34, CD31, ERG, p63, GFAP, S100, CK AE1/AE3, CK7 | 6/AND | |||
| De Ravin et al. 62 | 2022 | 1 | F | 81 | TP | SFT | CD34, CD99, ALDH1, bcl-2, CD99 | STAT6, S100, desmin | NA | |||
| Lee et al. 63 | 2022 | 1 | F | 33 | SP | Malignant SFT | CD34, STAT-6, FLI-1 | Pan-CK, CAM5.2, p63, S100, CD31, SMA, calponin | RT | 11/AND | ||
| Muñoz Guerra et al. 64 | 2022 | 1 | F | 37 | TP | SFT | CD34, bcl-2 | S100, CK | 12/AND | |||
| SFT: solitary fibrous tumour; HPC: haemangiopericytoma; TP: total parotidectomy; SP: superficial parotidectomy; ER: enucleoresection; STP: subtotal parotidectomy; NA: not available; DOD: dead of disease; AND: alive and without disease; AWD: alive with disease. | ||||||||||||
| Time (mean) | Sites | Histology | |
|---|---|---|---|
| Metastasis (3/6 = 50%) | 31.2 months | Lungs (3) muscles (1), liver (1), vertebrae (1), pelvis (1), retroperitoneal carcinomatosis (1) | Malignant HPC (3 + 1 uncertain case) |
| Recurrence (6/53 = 11.3%) | 24.2 months | Local recurrence | SFT (4) |
| Malignant HPC (1) | |||
| HPC (1) |
References
- Faur A, Buzaş R, Lăzărescu A. Current developments in diagnosis of salivary gland tumors: from structure to artificial intelligence. Life (Basel). 2024;14. doi:https://doi.org/10.3390/life14060727
- Fletcher C. 2013. WHO Classification of Tumours. International Agency for Research on Cancer; 2013.
- Stout A, Murray M. Hemangiopericytoma: a vascular tumor featuring Zimmermann’s pericytes. Ann Surg. 1942;116:26-33. doi:https://doi.org/10.1097/00000658-194207000-00004
- Chintale S, Katre M, Kirdak V. Hemangiopericytoma an incidental parotid swelling: a rare case report. Indian J Otolaryngol Head Neck Surg. 2022;74:2417-2421. doi:https://doi.org/10.1007/s12070-020-02204-1
- Batsakis J, Rice D. The pathology of head and neck tumors: vasoformative tumors, part 9A. Head Neck Surg. 1981;3:231-239. doi:https://doi.org/10.1002/hed.2890030311
- Smith S, Gooding W, Elkins M. Solitary fibrous tumors of the head and neck: a multi-institutional clinicopathologic study. Am J Surg Pathol. 2017;41:1642-1656. doi:https://doi.org/10.1097/PAS.0000000000000940
- Mosquera J, Fletcher C. Expanding the spectrum of malignant progression in solitary fibrous tumors: a study of 8 cases with a discrete anaplastic component – Is this dedifferentiated SFT?. Am J Surg Pathol. 2009;33:1314-1321. doi:https://doi.org/10.1097/pas.0b013e3181a6cd33
- Robinson D, Wu Y, Kalyana-Sundaram S. Identification of recurrent NAB2-STAT6 gene fusions in solitary fibrous tumor by integrative sequencing. Nat Genet. 2013;45:180-185. doi:https://doi.org/10.1038/ng.2509
- de Perrot M, Fischer S, Bründler M. Solitary fibrous tumors of the pleura. Ann Thorac Surg. 2002;74:285-293. doi:https://doi.org/10.1016/s0003-4975(01)03374-4
- Yoshida A, Tsuta K, Ohno M. STAT6 immunohistochemistry is helpful in the diagnosis of solitary fibrous tumors. Am J Surg Pathol. 2014;38:552-559. doi:https://doi.org/10.1097/PAS.0000000000000137
- Doyle L, Vivero M, Fletcher C. Nuclear expression of STAT6 distinguishes solitary fibrous tumor from histologic mimics. Mod Pathol. 2014;27:390-395. doi:https://doi.org/10.1038/modpathol.2013.164
- Ganly I, Patel S, Stambuk H. Solitary fibrous tumors of the head and neck: a clinicopathologic and radiologic review. Arch Otolaryngol Head Neck Surg. 2006;132:517-525. doi:https://doi.org/10.1001/archotol.132.5.517
- Norton S, Clark S, Sheehan A. Solitary fibrous tumour of the diaphragm. J Cardiovasc Surg (Torino). 1997;38:685-686.
- Chick J, Chauhan N, Madan R. Solitary fibrous tumors of the thorax: nomenclature, epidemiology, radiologic and pathologic findings, differential diagnoses, and management. AJR Am J Roentgenol. 2013;200:W238-W248. doi:https://doi.org/10.2214/AJR.11.8430
- Wang K, Mei F, Wu S. Hemangiopericytoma: incidence, treatment, and prognosis analysis based on SEER database. Biomed Res Int. 2020;2020. doi:https://doi.org/10.1155/2020/2468320
- Baldi G, Stacchiotti S, Mauro V. Solitary fibrous tumor of all sites: outcome of late recurrences in 14 patients. Clin Sarcoma Res. 2013;3. doi:https://doi.org/10.1186/2045-3329-3-4
- Page M, McKenzie J, Bossuyt P. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372. doi:https://doi.org/10.1136/bmj.n71
- Neal T, Starke W. Hemangiopericytoma of the parotid gland: a case report with autopsy. Laryngoscope. 1973;83:1953-1958. doi:https://doi.org/10.1288/00005537-197312000-00006
- Katz A. Unusual lesions of the parotid gland. J Surg Oncol. 1975;7:219-235. doi:https://doi.org/10.1002/jso.2930070308
- Massarelli G, Tanda F, Fois V. Haemangiopericytoma of the parotid gland. Report of a case and review of the literature. Virchows Arch A Pathol Anat Histol. 1980;386:81-89. doi:https://doi.org/10.1007/BF00432646
- Katsantonis G, Friedman W, Rosenblum B. The surgical management of advanced malignancies of the parotid gland. Otolaryngol Head Neck Surg. 1989;101:633-640. doi:https://doi.org/10.1177/019459988910100604
- Carrillo R, Rodriquez-Peralto J, Batsakis J. Primary haemangiopericytomas of the parotid gland. J Laryngol Otol. 1992;106:659-661. doi:https://doi.org/10.1017/s0022215100120468
- Lee G, Wong D, Chang R. Hemangiopericytoma of the parotid gland: report of case. J Oral Maxillofac Surg. 1992;50:1329-1332. doi:https://doi.org/10.1016/0278-2391(92)90238-u
- Whittam D, Hellier W. Haemangiopericytoma of the parotid salivary gland: report of a case with literature review. J Laryngol Otol. 1993;107:1159-1162. doi:https://doi.org/10.1017/s0022215100125551
- Hanau C, Miettinen M. Solitary fibrous tumor: histological and immunohistochemical spectrum of benign and malignant variants presenting at different sites. Hum Pathol. 1995;26:440-449. doi:https://doi.org/10.1016/0046-8177(95)90147-7
- Ferreiro J, Nascimento A. Solitary fibrous tumour of the major salivary glands. Histopathology. 1996;28:261-264. doi:https://doi.org/10.1046/j.1365-2559.1996.d01-419.x
- Sato J, Asakura K, Yokoyama Y. Solitary fibrous tumor of the parotid gland extending to the parapharyngeal space. Eur Arch Otorhinolaryngol. 1998;255:18-21. doi:https://doi.org/10.1007/s004050050015
- Sawh R, Lele S, Borkowski J. Fine-needle aspiration cytology of hemangiopericytoma: report of two cases. Diagn Cytopathol. 2000;23:187-191. doi:https://doi.org/10.1002/1097-0339(200009)23:3<187::aid-dc9>3.0.co;2-y
- Mohammed K, Harbourne G, Walsh M. Solitary fibrous tumour of the parotid gland. J Laryngol Otol. 2001;115:831-832. doi:https://doi.org/10.1258/0022215011909099
- Kumagai M, Suzuki H, Takahashi E. A case of solitary fibrous tumor of the parotid gland: review of the literature. Tohoku J Exp Med. 2002;198:41-46. doi:https://doi.org/10.1620/tjem.198.41
- Sikes J, Ghali G, Veillon D. Parotid mass in a 62-year-old man. J Oral Maxillofac Surg. 2003;61:1073-1077. doi:https://doi.org/10.1016/s0278-2391(03)00320-3
- Thompson M, Cheng L, Stewart J. A paediatric case of a solitary fibrous tumour of the parotid gland. Int J Pediatr Otorhinolaryngol. 2004;68:481-487. doi:https://doi.org/10.1016/j.ijporl.2003.10.023
- Gerhard R, Fregnani E, Falzoni R. Cytologic features of solitary fibrous tumor of the parotid gland. A case report. Acta Cytol. 2004;48:402-406. doi:https://doi.org/10.1159/000326393
- Wiriosuparto S, Krassilnik N, Bhuta S. Solitary fibrous tumor: report of a case with an unusual presentation as a spindle cell parotid neoplasm. Acta Cytol. 2005;49:309-313. doi:https://doi.org/10.1159/000326154
- Sreetharan S, Prepageran N. Benign fibrous tumour of the parotid gland. Asian J Surg. 2005;28:45-47. doi:https://doi.org/10.1016/S1015-9584(09)60258-X
- Balaji R, Khoo J, Sittampalam K. CT imaging of malignant metastatic hemangiopericytoma of the parotid gland with histopathological correlation. Cancer Imaging. 2008;8:186-190. doi:https://doi.org/10.1102/1470-7330.2008.0030
- Cho K, Ro J, Choi J. Mesenchymal neoplasms of the major salivary glands: clinicopathological features of 18 cases. Eur Arch Otorhinolaryngol. 2008;265:S47-S56. doi:https://doi.org/10.1007/s00405-007-0488-5
- Manglik N, Patil S, Reed M. Solitary fibrous tumour of the parotid gland. Pathology. 2008;40:89-91. doi:https://doi.org/10.1080/00313020701716334
- Bauer J, Miklos A, Thompson L. Parotid gland solitary fibrous tumor: a case report and clinicopathologic review of 22 cases from the literature. Head Neck Pathol. 2012;6:21-31. doi:https://doi.org/10.1007/s12105-011-0305-8
- Nowlin R, Kokot N. Hemangiopericytoma of the parotid: a case report. Laryngoscope. 2011;121. doi:https://doi.org/10.1002/lary.22016
- Iyengar J, Atmaram M, Neeli D. Solitary fibrous tumor presenting as a mass in the parotid gland. Indian J Pathol Microbiol. 2011;54:612-613. doi:https://doi.org/10.4103/0377-4929.85114
- Cristofaro M, Allegra E, Giudice M. Two new localizations of solitary fibrous tumor in the italian population: parotid gland and oral cavity-review of the literature. J Oral Maxillofac Surg. 2012;70:2360-2367. doi:https://doi.org/10.1016/j.joms.2011.10.023
- Chen D, Xuan J, Sun M. Lipomatous hemangiopericytoma (adipocytic variant of solitary fibrous tumor) of the parotid gland: a case report and review of the literature. Oncol Lett. 2013;6:1380-1382. doi:https://doi.org/10.3892/ol.2013.1538
- Sousa A, Souto G, Sousa I. Solitary fibrous tumor of the parotid gland: case report. J Clin Exp Dent. 2013;5:E208-E211. doi:https://doi.org/10.4317/jced.51103
- Apuhan T, Iwenofu H, Özer E. Solitary fibrous tumor of the accessory parotid gland: a unique case. Kulak Burun Bogaz Ihtis Derg. 2013;23:239-241. doi:https://doi.org/10.5606/kbbihtisas.2013.42714
- Liu Y, Li K, Shi H. Solitary fibrous tumours in the extracranial head and neck region: correlation of CT and MR features with pathologic findings. Radiol Med. 2014;119:910-919. doi:https://doi.org/10.1007/s11547-014-0409-9
- Chis O, Albu S. Giant solitary fibrous tumor of the parotid gland. Case Rep Med. 2014;2014. doi:https://doi.org/10.1155/2014/950712
- Alonso-Rodríguez E, González-Otero T, Castro-Calvo A. Parotid gland solitary fibrous tumor with mandibular bone destruction and aggressive behavior. J Clin Exp Dent. 2014;6:E299-E302. doi:https://doi.org/10.4317/jced.51256
- Künzel J, Hainz M, Ziebart T. Head and neck solitary fibrous tumors: a rare and challenging entity. Eur Arch Otorhinolaryngol. 2016;273:1589-1598. doi:https://doi.org/10.1007/s00405-015-3670-1
- Yu R, Rebello R. Solitary fibrous tumor of the parotid gland: a case report. Iran J Otorhinolaryngol. 2015;27:401-405.
- Gervasio C, Cerutti M, Dalmasso G. A rare case of solitary fibrous tumor of parotid gland: diagnostic work-up and treatment. Otorinolaringologia. 2017;67:116-119. doi:https://doi.org/10.23736/S0392-6621.17.02129-4
- Rais M, Kessab A, Sayad Z. Solitary fibrous tumor occurring in the parotid gland: a case report. BMC Clin Pathol. 2017;17. doi:https://doi.org/10.1186/s12907-017-0062-z
- Ralli M, Marwah N, Agarwal M. Solitary fibrous tumor of the parotid gland in a young female: a rare case report and review of the literature. MEJC. 2018;9:65-69. doi:https://doi.org/10.30476/mejc.2018.42094
- Lim D, Tan T, Tan J. Solitary fibrous tumour of the parotid gland: a case report and a 15-year literature review. AME Case Rep. 2019;3. doi:https://doi.org/10.21037/acr.2019.04.05
- Romano N, Ferrari A, Moroni M. Solitary fibrous tumor of the deep parotid gland. Ear Nose Throat J. 2022;101:NP291-NP293. doi:https://doi.org/10.1177/0145561320966068
- Velez Torres J, Duarte E, Diaz-Perez J. Mesenchymal neoplasms of salivary glands: a clinicopathologic study of 68 cases. Head Neck Pathol. 2022;16:353-365. doi:https://doi.org/10.1007/s12105-021-01360-2
- Vitale A, Paolella C, Rossi G. Recurrent fibrous solitary tumor of the parotid gland with satellite location: imaging, clinical and histological findings of rare entity. Radiol Case Rep. 2021;16:2924-2928. doi:https://doi.org/10.1016/j.radcr.2021.07.007
- Li Y, Li C, Liu Z. Dumbbell-shaped solitary fibrous tumor in the parapharyngeal space: a case report. World J Clin Cases. 2021;9:1204-1209. doi:https://doi.org/10.12998/wjcc.v9.i5.1204
- Abdulhaleem K, Dababo M, Othman E. Parotid gland solitary fibrous tumor presenting as a long duration mass: a case report. Case Rep Pathol. 2022;2022. doi:https://doi.org/10.1155/2022/2097634
- Goker F, Mazzucato C, Maggioni M. Solitary fibrous tumor of parotid gland: a case report and short review of literature. Eur Rev Med Pharmacol Sci. 2022;26:45-50. doi:https://doi.org/10.26355/eurrev_202212_30794
- Boschetti C, Vitagliano R, Imola G. Solitary extrapleural fibrous tumor in salivary glands: our experience-case series and literature review. Diagnostics (Basel). 2022;12. doi:https://doi.org/10.3390/diagnostics12112688
- De Ravin E, Marrero Berrios J, Rajasekaran K. Parotid mass in an 81-year-old woman. JAMA Otolaryngol Head Neck Surg. 2022;148:981-982. doi:https://doi.org/10.1001/jamaoto.2022.2249
- Lee C, Liu K, Huang S. A dedifferentiated solitary fibrous tumor of the parotid gland: a case report with cytopathologic findings and review of the literature. Diagn Pathol. 2019;14. doi:https://doi.org/10.1186/s13000-019-0792-6
- Guerra M, Amat C, Campo F. Solitary fibrous tumor of the parotid gland: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002;94:78-82. doi:https://doi.org/10.1067/moe.2002.121990
- Mortelé B, Lemmerling M, Seynaeve P. Hemangiopericytoma of the parotid gland: CT and MR features. Eur Radiol. 2001;11:1073-1075. doi:https://doi.org/10.1007/s003300000463
- Lee D, Yoon T, Lee J. Clinical features of pediatric parotid tumors: 10-year experience of a single institute. Acta Otolaryngol. 2013;133:1213-1218. doi:https://doi.org/10.3109/00016489.2013.822554
- Zamani M, Grønhøj C, Schmidt Jensen J. Survival and characteristics of pediatric salivary gland cancer: a systematic review and meta-analysis. Pediatr Blood Cancer. 2019;66. doi:https://doi.org/10.1002/pbc.27543
- Younes A, Taher M, Sidhom I. Parotid gland masses: outcomes in the pediatric age group. J Egypt Natl Canc Inst. 2023;35. doi:https://doi.org/10.1186/s43046-023-00161-8
- Liberale C, Rozell-Shannon L, Moneghini L. Stop calling me cavernous hemangioma! A literature review on misdiagnosed bony vascular anomalies. J Invest Surg. 2022;35:141-150. doi:https://doi.org/10.1080/08941939.2020.1824041
Downloads
License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright
Copyright (c) 2026 Società Italiana di Otorinolaringoiatria e chirurgia cervico facciale
How to Cite
- Abstract viewed - 626 times
- PDF downloaded - 133 times




