Reviews
Vol. 46: Issue 1 - February 2026
Regional and distant metastases from laryngeal chondrosarcoma: a systematic review
Summary
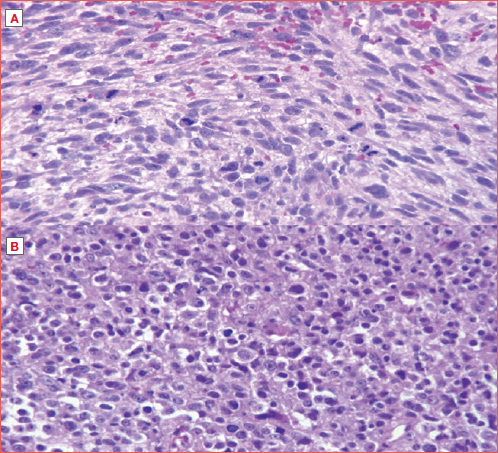
Objective. Laryngeal chondrosarcomas (LCS) are rare malignancies, constituting approximately 0.5% of all primary laryngeal tumours. These predominantly slow-growing, low-grade tumors can sometimes exhibit a more aggressive clinical course with higher rates of local recurrence and possible regional and/or distant metastases. The true occurrence of metastatic LCS is not well defined in the literature due to limited and scattered data. This systematic review aims to analyse the metastatic behaviour of LCS, focusing on patterns of spread, therapeutic options, and clinical outcomes.
Methods. A systematic review was conducted according to the PRISMA guidelines searching on PubMed, Web of Science, and Scopus databases. Studies included cases of LCS with regional and/or distant metastasis.
Results. A total of 44 articles describing 53 cases of metastatic LCS were included. The mean age of patients was 63.5 years. Aggressive subtypes of LCS (high grade, dedifferentiated, and myxoid variants) accounted for 54.3% of metastatic cases. Distant metastases were observed in 71.7% of patients, with lungs being the most common site. Regional lymph nodes involvement was documented in 37.7% of cases. Surgery was the primary treatment, with total laryngectomy being the most common approach. Radiotherapy was administered as adjuvant treatment in 24.5% of patients. At last follow-up, 49% of patients had died from the disease, and 9.4% had persistent disease.
Conclusions. Although LCS is typically indolent, patients with metastatic disease present significant clinical challenges. The true metastatic rate remains uncertain due to the scattered distribution of data. Aggressive subtypes of LCS exhibit a greater propensity for metastasis, underscoring the need for closer surveillance and individualised treatment strategies.
Introduction
Laryngeal chondrosarcomas (LCS) are rare tumours accounting for about 0.5% of all laryngeal primary tumours, even though they represent the most frequent non-epithelial malignancy of this organ 1. These tumours predominantly affect the cricoid cartilage and are more commonly observed in patients between the age of 50 and 70 years, but may also occur in younger subjects 2. Treatment is mainly based on laryngeal-sparing surgical excision, which can be achieved endoscopically via transoral laser microsurgery (TLM) or by a number of open-neck conservative techniques, reserving upfront total laryngectomy (TL) to quite advanced, recurrent, or aggressive lesions, affecting older patients who are no more amenable of organ preservation strategies 3-8. Chemotherapy (CHT) and radiotherapy (RT) are not recommended as upfront treatments 9, although successful definitive proton therapy has been recently reported 10.
Generally, LCS are slow-growing tumours with relatively low malignant potential. However, high-grade (G3), dedifferentiated (DD), and myxoid (MY) LCS are considered more aggressive subtypes due to their higher rates of local recurrence, distant metastases (DM), and relatively poor prognosis 8 (Cover figure). Metastases from LCS have been described in the literature in up to 10% of cases, although some studies reported a lower incidence of 1.9% 1. Metastatic patterns of LCS are typically haematogenous with lung and bone being the most prevalent metastatic sites. Considering the cervical lymph nodes, regional metastases are rare, and neck dissection must be reserved for cases in which diagnostic imaging suggests clinical lymph node(s) involvement 11.
Limited and scattered data, mostly in form of case reports, regarding metastatic LCS are available in the literature. The aim of this review is therefore to provide a comprehensive analysis of the metastatic behaviour of LCS, with a focus on its patterns of spread, therapeutic options, and clinical outcomes.
Materials and methods
Search strategy
This systematic review was conducted following the guidelines outlined in the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) checklist and statement 12. A thorough literature search was performed across PubMed, Web of Science, and Scopus in February 2025 using the query: “(Chondrosarcoma or chondrosarcomas or chondrosarcomatoid or sarcoma or sarcomas) and (larynx or laryngeal) and (metastasis or lymph nodes or lymph node or node or nodes or lymphatic or metastatic or neck metastasis or regional metastasis or neck or distant metastasis)”.
Selection criteria
Original research articles in English and Italian, published until February 2025 that reported cases of LCS with regional involvement and/or DM at diagnosis or during follow-up were included. Studies were excluded if they involved non-human subjects, or lacked specific data from composite series or were reviews or articles without full-text availability. Two independent researchers (CM and AF) screened all titles, reviewed abstracts, and excluded studies that did not meet the inclusion criteria. Full-text articles were then assessed to determine their eligibility based on the predefined criteria. In cases of disagreement, a third investigator (CP) acted as a mediator to reach a consensus. Additionally, references of all selected articles were examined to identify further relevant studies.
Data extraction
The data extracted from each included study comprised the authors’ names, year of publication, journal, as well as number of cases, and gender and age of patients at presentation. Additionally, we gathered information on tumour characteristics (site of origin and histologic subtype), presence and site of regional and/or distant spread at diagnosis or during follow-up, therapeutic approach, length of follow-up, and status at the last control. Two authors (CM and AF) independently reviewed and extracted data from the selected articles, compiling the information into a dedicated Microsoft Excel database. This article is based on previously published research and does not involve any new studies with human participants or animals conducted by the authors.
Results
General findings
The initial search returned 4,581 articles. All details about identification, screening, and inclusion/exclusion criteria are reported in Figure 1. A total of 44 articles 1,5,11,13-53, case reports and case series, were selected. In total, 53 patients with metastatic LCS were included in the present systematic review.
Demographics and tumour characteristics
Demographics were stated for almost all patients, except for 15 cases 5,14,24,34,42,46,47,50,51. Mean age of our population was 63.5 years (range, 38-82). When reported, the male to female ratio was 2 to 1, counting 26 males and 12 females. Excluding 16 cases (30.2%) 14,22,28,34,38,41,42,46,47,50,51, whose exact origin was not specified, the cricoid was the most common site of origin of LCS (n = 25, 67.6%) 1,5,13,15,17-21,23,26,30,31,37,39,40,43,45,49,52,53. Among these, in one case (2.7%) each, cricoid was involved with the thyroid 45 and the arytenoid 53, respectively. Rarely, the primary site was the epiglottis (n = 3, 8.1%) 24,25,27, arytenoid (n = 1, 2.7%) 32, or corniculate cartilage (n = 1, 2.7%)16. Histological subtypes were reported in 35 cases 1,5,11,17-19,25-33,35-40,43-45,48-50,52,53. Only 5 patients (14.3%) had a well-differentiated (G1) LCS 21,26,48,50,52, while nearly half had an aggressive subtype ([G3, DD, or MY], n = 19, 54.3%) 1,17-19,25,27,28,30-32,35-38,43,44,53. Details are reported in Table I.
Metastatic patterns
All LCS selected had distant and/or regional metastasis at diagnosis or during follow-up. Thirty-eight patients (71.7%) had LCS with DM 1,11,13,15,17,19-24,26-28,31-36,38-44,46,48-53. In details, when specified, DM were detected at diagnosis and during follow-up in 4 (7.5%) 17,39,46,52 and 28 (52.9%) patients 1,5,11,13,15,20-23,26,28,31-33,35,36,38,40-44,48-50,53, respectively. During follow-up, when available, the mean time of DM detection was 23.8 months (range, 3-84). The most common sites of DM were lungs, described in 29 patients (54.7%) 1,5,11,15,17,19,21-24,26-28,31,32,35,36,38-41,44,48,50,52,53. Other rare sites were bone (n = 4, 7.5%) 31,39,41,43, brain (n = 2, 3.8%) 32,34, kidney (n = 1, 1.9%) 19, and liver (n = 1, 1.9%) 48. Regional lymph nodes involvement was reported in 20 patients (37.7%) 14-16,18,19,25-27,29-31,45-47,49,53. It was observed in 6 (11.3%) at diagnosis 18,25,45,46 and in 7 (13.2%) during follow-up 15,16,30,31,49,53 with a mean time of 12.8 months (range, 5-24). Additional details are reported in Table I.
Treatment and follow-up
Surgery was performed in nearly all patients (77.4%), except for 11 whose treatment was not specified 14,24,42,46,47,50,51, and one (1.9%) who was managed with palliative care alone 50. The most common treatment was TL performed in 37.7% (n = 20) of cases as primary treatment 1,11,15,16,21,25,26,28,29,31,32,35-37,40,43-45, and in 20.8% (n = 11) 5,13,18,19,23,30,31,33,48,53 as secondary treatment after failure of previous conservative surgical techniques. Conservative surgical approaches, including partial laryngectomies or local resections, were performed in 6 (11.3%) patients 17,20,22,27,41,49. Thirteen (24.5%) received adjuvant RT after surgery 1,11,16,18,28,35,35,36,38,43,44,48,49, while 3 patients underwent RT with palliative intent 22,31,39. Mean follow-up, when available, was 50.2 months (range, 0-360 months). At the last follow-up, more than half of our population had died (n = 28, 52.8%): 26 (49%) died of disease (DOD) 1,11,15-21,24,28,32-37,39-41,43,44,48,52,53 and 2 (3.8%) from other causes (DOC) 30,31. Five patients (9.4%) were alive with disease (AWD) 5,23,31,49. All data are reported in Table I.
Discussion
LCS is the most frequent sarcoma of the larynx 2. Compared to chondrosarcomas of other sites of the body, laryngeal ones tend to be low-grade and less aggressive 54, even though it is not clear if this depends on an intrinsic biologic behaviour of LCS or, more probably, from their well-confined position of the larynx within the body and the consequent relatively early diagnosis due to dysphonia and dyspnoea. The prognosis of LCS is, in fact, generally favourable, due to its usual indolence and low propensity to metastatic diffusion 55. Indeed, surgeons have seemingly prioritised preserving the larynx over reducing the recurrence rate, accepting a higher likelihood of tumour persistence/recurrence due to its low impact on overall and disease-specific survivals 56. However, in rare cases, these tumours may progress to regional and DM. One of the largest series, involving 111 LCS diagnosed between 1970 and 1997 1, reported a metastatic rate of 1.9%. However, still today, the true incidence of metastatic LCS remains unreliable and not well defined. Indeed, the reported percentages of metastases from LCS described in up to 10% of cases hold little significance, as they are often based on small case series or rely on data from other authors. Our systematic review provides an in-depth analysis of the metastatic behaviour of LCS, with a focus on its patterns of spread and clinical outcomes.
General findings
A total of 44 articles and 53 metastatic LCS were included in the present review 1,5,11,13-53. In the article selection process, we excluded the case reported by Neel and Unni 57, as the authors did not conclusively determine whether the tumour found in the cervical spine was a metastasis or a further metachronous primary tumour. Moreover, it was not considered a metastasis in the more recent reviews by Lewis et al. 58 and Nakayama et al. 31.
Demographics and tumour characteristics
While the location of primary LCS is one of the most important determinants for recurrence and organ preservation, for metastatic potential no clear risk factors are known. Our population had a mean age of 63.5 years (range, 38-82) with a male predominance, and the most frequent site of origin of the tumour was at the level of the cricoid, as described for LCS series in general. No differences in terms of demographics between metastatic and non-metastatic LCS were found. According to the histopathological subtypes, G1 LCS accounted for only 14.3% of metastatic cases 21,26,48,50,52, while more than half (54.3%) of the metastatic tumours were G3, DD, and MY 1,17-19,25,27,28,30-32,35-38,43,44,53. These findings reinforce the prognostic significance of histological grading of LCS, suggesting that aggressive subtypes necessitate closer monitoring and potentially more extensive initial treatment. Although rarely, G1 LCS can sometimes present metastasis. However, determining whether these cases were truly G1 is challenging due to the well-documented difficulties and subjectivity with high intra- and inter-observer variability in the histopathological evaluation of chondrosarcoma aggressiveness. In fact, assessing cartilaginous tumours may often be complex and time-consuming, particularly in terms of classification and grading 21.
Metastatic pattern
In our population, all 53 cases of LCS had distant and/or regional metastasis at diagnosis or during follow-up. DM were more frequent than regional involvement (71.7% vs 37.7%, respectively). The timing of detection was not always specified, but, when available, regional metastasis were detected half at the diagnosis and half during follow-up with a mean time of 12.8 months. Interestingly, DM were detected at initial diagnosis in only 7.5% of cases, whereas the majority of patients (52.9%) developed them during follow-up (mean time of detection, 23.8 months), emphasising the necessity for long-term surveillance. The most common sites of DM were lungs, and rarely bones, brain, kidney or liver. In 1993, Nakayama et al. 31 published a review of the literature regarding regional metastasis and/or DM of LCS that revealed a total of 20 cases. Lavertu et al. 59 highlighted DM in 5 of the 35 cricoid cases (14%), while Thompson and Gannon 1 reported a series of 111 LCS and demonstrated that local and DM rates were exceedingly low, around 1.9%. However, accurate estimation of the incidence of metastatic LCS is seriously hampered by several issues. For instance, there is a lack of knowledge of the true total number of chondrosarcomas documented in the literature, and cases of LCS are likely more frequent than reported, as they were often classified under the broader category of cartilaginous tumours and historically misdiagnosed as chondromas 2. The reported 10% metastasis rate in LCS is unreliable, despite being erroneously cited by several authors. Based on our data, the metastatic rate of 1.9% reported by Thompson and Gannon 1 appears to be the most reliable estimate, even though the lack of large series or prospective studies make it impossible to define a more precise rate.
Treatment and follow-up
Surgical intervention was the primary treatment modality, with TL performed in 37.7% as the initial approach 1,11,15,16,21,25,26,28,29,31,32,35-37,40,43-45 and in 20.8% after failure of conservative surgery 5,13,18,19,23,30,31,33,48,53. Partial laryngectomies or local resections were attempted in 11.3% 17,20,22,27,41,49 of cases, reflecting a low trend toward organ preservation in these cases. The high rate of secondary TL highlights the potential limitations of conservative approaches, particularly in aggressive tumour subtypes. According to the literature, G3 LCS, and especially DD, should be treated as separate entities from conventional chondrosarcomas because of their proclivity for more aggressive behaviours 8,26.
Thirteen (24.5%) patients had adjuvant RT after surgery 1,11,16,18,28,35,35,36,38,43,44,48,49, while 3 underwent RT with palliative intent 22,31,39. Historically, LCS were considered as radioresistant tumours, although evidence in the literature has shown that these lesions can no longer be classified as radioresistant entities. RT could be performed in case of unfeasible surgical resection, recurrent tumours or in the adjuvant setting, in case of doubt of inadequate surgical resection 22.
Overall, the prognosis of patients with LCS is generally favourable, with 5-year survival rates exceeding 80% 56. In our cohort, survival outcome was poor, revealing a 49% disease-specific mortality rate with a mean follow-up of 55.1 months. Disease persistence was noted in 5 patients 5,23,31,49, further underscoring the challenges in managing metastatic LCS. The occurrence of metastases significantly worsens outcomes, with survival rates dropping considerably. This emphasises the need for a more radical primary approach for specific subtypes of LCS, individualised treatment strategies, long-term follow-up, and further research into novel therapeutic options. Indeed, even if regional lymph node metastases and DM are uncommon, when present they may represent significant clinical challenges. Understanding the factors that contribute to the metastatic behaviour of LCS is crucial for risk stratification and management. Future research should focus on elucidating the molecular mechanisms underlying metastasis in LCS to develop targeted therapeutic approaches.
Conclusions
LCS is a rare tumour with generally low aggressiveness and favourable prognosis. However, it can sometimes exhibit a higher risk of local recurrence and regional and/or DM, significantly worsening outcomes. Our systematic review confirms that metastases from LCS are uncommon, with an incidence that remains difficult to determine due to the scarcity of large-scale studies. DM, primarily affecting the lungs, are more frequent than regional lymph nodes involvement and often arise several months or years after initial diagnosis, highlighting the need for extended follow-up. A better understanding of metastatic risk factors, individualised treatment approaches, and long-term follow-up strategies are essential. Future multicentre and prospective studies will be crucial in refining the management and therapeutic options for this rare but clinically important disease.
Conflict of interest statement
The authors declare no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author contributions
CM, AF: article selection and data extraction; CP: mediator to reach a consensus; CP, AF, CM: study design, review drafting, and critical revision.
Ethical consideration
No formal ethics committee approval was required for this article as it is based on already published clinical data from other studies available in the literature.
History
Received: April 27, 2025
Accepted: May 29, 2025
Figures and tables
Figure 1. Selection process of original papers published until February 2025, describing cases of metastatic LCS.
| First author | Year | No. of cases | Age (y) | Gender | Site | Histological subtype | Treatment | N+/M+ | N+ AT diagnosis | M+ AT diagnosis | Status FU | Mean FU (m) | N+ during FU | Site | Time (m) | M+ during FU | Site | Time (m) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ungerecht 13 | 1951 | 1 | 62 | M | C | NR | Laryngofissure and TL 7 m later | M+ | NR | NR | NR | NR | NR | NR | NR | YES | LUNGS | NR | |
| Frenzel 14 | 1953 | 1 | NR | NR | NR | NR | NR | C. NODES | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | |
| Sirota 15 | 1958 | 1 | 65 | M | C | NR | TL | C. NODES, LUNGS, RENAL ARTERY | NR | NR | DOD | 28 | YES | C. NODES | NR | YES | LUNGS AND RENAL ARTERY TUMOUR EMBOLUS | 24 | |
| Bronzini 16 | 1959 | 1 | 72 | M | COR | NR | TL + RT; left RND +RT 7 m later | C. NODES, WIDESPREAD | NR | NR | DOD | 18 | YES | C. NODES | NR | NO | None | 0 | |
| Ghalib *17 | 1969 | 1 | 74 | F | C | G2-G3 | TT + excision soft tissue mass; tracheotomy 1 d later | LUNGS | NO | YES | DOD | 30 | NR | NR | NR | NR | NR | NR | |
| Al-Saleem 18 | 1970 | 1 | 70 | F | C | G3/DD | Several endoscopic resections; TL + RT for N+ 1 m later | C. NODES | YES | NO | DOD | 8 | NR | NR | NR | NR | NR | NR | |
| Huizenga and Balogh *19 | 1970 | 1 | 68 | M | C | G3 | Laryngofissure; TL 5 y later; local excision 3 y later; RT 9 y later | LUNG, KIDNEY, AND NECK | NR | NR | DOD | NR | NR | NR | NR | NR | NR | NR | |
| Hyams and Rabuzzi 20 | 1970 | 1 | 71 | F | C | NR | 2 local resection; tracheotomy for recurrence 23 y later; RT | SUBCUTANEOUS THORACIC AND ABDOMINAL NODULES | NO | NO | DOD | 360 | NO | None | 0 | YES | SUBCUTANEOUS THORACIC AND ABDOMINAL NODULES | NR | |
| Hellquist 21 | 1979 | 1 | 65 | F | C | G1 | Tracheotomy; TL 1 m later | LUNGS, SPLEEN | NO | NO | DOD | 42 | NO | None | 0 | YES | LUNGS, SPLEEN | 36 | |
| Harwood 22 | 1980 | 1 | 75 | M | NR | NR | Excision; palliative RT for lung metastases | LUNGS | NR | NR | NR | NR | NR | NR | NR | YES | LUNGS | NR | |
| Ferlito 11 | 1984 | 1 | 74 | M | T | G2 | TL + RT | LUNGS | NO | NO | DOD | 30 | NO | None | 0 | YES | LUNGS | 12 | |
| Escher 23 | 1984 | 1 | 53 | M | C | NR | Laryngofissure; multiple resections 4, 5, and 6 y later; then after TL 1 y later | LUNGS | NR | NR | AWD | 138 | NR | NR | NR | YES | LUNGS | NR | |
| Gray 24 | 1984 | 1 | NR | NR | E | NR | NR | LUNGS | NR | NR | DOD | Fulminating fatal outcome | NR | NR | NR | NR | NR | NR | |
| Kasanzew 25 | 1988 | 1 | 46 | F | E | G3 | Tracheotomy followed by TL + bilateral VI level dissection; left RND | PARALAYNGEAL NODES | YES | NO | NED | 6 | NO | None | 0 | NO | None | 0 | |
| Hakky 26 | 1989 | 1 | 56 | M | C | G1 | TL + ND | C. NODES, LUNGS | NR | NR | NED | 36 | NR | NR | NR | YES | LUNGS | 36 | |
| Jacobs 27 | 1989 | 1 | 52 | M | E | G2/G3 | PL + right mRND; segmental lobectomy 3 y later | C. NODES, LUNGS | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | |
| Nicolai 28 | 1990 | 1 | 74 | M | NR | DD | TL + RT | LUNGS | NO | NO | DOD | 30 | NO | None | 0 | YES | LUNGS | 12 | |
| Glaubiger 29 | 1991 | 1 | 57 | M | T | G2 | TL + PP + TT + left mRND + right RND | C. AND MEDIASTINAL NODES | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | |
| Brandwein 30 | 1992 | 1 | 69 | M | C | DD | PL; TLM; TL; tracheal resection; RT | C. NODES | NO | NO | DOC | 216 | YES | C. NODES | 18 | NO | None | 0 | |
| Nakayama 31 | 1993 | 2 | 58 | F | C | DD | TL; excision of stomal recurrence 4 m later; ND 1 m later; excision of recurrence 5 y later | C. NODES | NO | NO | DOC | 168 | YES | C. NODES | 5 (4 regional recurrences during subsequent 8 y) | NO | None | 0 | |
| 62 | F | C | DD | Tracheotomy + laryngofissure; TL + excision distal clavicle 21 m later; palliative RT (bone) 3 m later; further RT on chest wall later on | CLAVICLE, C. NODES, LUNGS, BONES (VERTEBRAL), LEFT ANTERIOR CHEST WALL | NO | NO | AWD | NR | YES | C. NODES | 24 | YES | DISTAL CLAVICLE, LUNGS AND BONES, LEFT ANTERIOR CHEST WALL | 21 (distal clavicle), 24 (lungs and vertebral) | ||||
| Moran 32 | 1993 | 1 | 61 | F | ARY | MY | TL | LUNGS, BRAIN | NO | NO | DOD | 15 | NO | None | 0 | YES | LUNGS, BRAIN | 15 | |
| Fichera 33 | 1995 | 1 | 60 | F | T | G2 | Laryngofissure; TL 6 y later | M+ (PLURIVISCERAL METASTASIS) | NO | NO | DOD | 6 | NO | None | 0 | YES | PLURIVISCERAL | 6 | |
| Lippert 34 | 1997 | 1 | NR | NR | NR | NR | Surgery not otherwise specified | BRAIN | NR | NR | DOD | 3 | NR | NR | NR | NR | NR | NR | |
| Thompson 1 | 2002 | 2 | 66 | M | C | G2 | TL; RT | LUNGS | NO | NO | DOD | 20.4 | NO | None | 0 | YES | LUNGS | NR | |
| 62 | M | C | MY | TL; CRT | LUNGS | NO | NO | DOD | 19.2 | NO | None | 0 | YES | LUNGS | NR | ||||
| Jones 35 | 2003 | 1 | 72 | M | T | G2/G3 | TL + RT | LUNGS | NO | NO | DOD | 12 | NO | None | 0 | YES | LUNGS | NR | |
| Rinaggio 36 | 2004 | 1 | 60 | M | T | DD | Debulking and TL + TT + bilateral ND 2 w later + RT | LUNGS | NO | NO | DOD | 3 | NO | None | 0 | YES | LUNGS | 3 | |
| Casiraghi 37 | 2004 | 2 | 61 | M | C | DD | TL + ND | NR | NR | NR | NED | 60 | NR | NR | NR | NR | NR | NR | |
| 58 | M | C | DD | TL | NR | NR | NR | DOD | 24 | NR | NR | NR | NR | NR | NR | ||||
| Goda 38 | 2011 | 1 | 38 | M | NR | G3 | Surgery not otherwise specified + RT | LUNGS | NO | NO | NED | 119 | NO | None | 0 | YES | LUNGS | 12 | |
| Böscke 39 | 2012 | 1 | 81 | F | C | G2 | Tracheotomy + palliative RT of neck, shoulder, and mediastinum | LUNGS, HUMERUS | NO | YES | DOD | 2.5 | NR | NR | NR | NR | NR | NR | |
| Domínguez-Durán 40 | 2014 | 1 | 60 | M | C | G2 | TL; CHT 1 y later | LUNGS, SKIN | NO | NO | DOD | 35 | NO | None | 0 | YES | LUNGS, SKIN | 12 (LUNGS), 30 (SKIN) | |
| Leong 41 | 2014 | 1 | 39 | F | NR | NR | PL + ND + RFFF | LUNGS AND PELVIS | NO | NO | DOD | 25 | NO | None | 0 | YES | LUNGS, PELVIS | 25 | |
| Dubal 42 | 2014 | 1 | NR | NR | NR | NR | NR | M+ | NR | NR | NR | NR | NR | NR | NR | YES | NR | NR | |
| Magliocca 43 | 2017 | 1 | 76 | F | C | DD | TL + RT; palliative CHT 3 m later | SOFT TISSUE, BONES | NO | NO | DOD | 8 | NO | None | 0 | YES | SOFT TISSUES, BONES | 3 | |
| Lame 44 | 2017 | 1 | 65 | M | T | G3 | TL + bilateral ND + right thyroid lobectomy + RT | LUNGS | NO | NO | DOD | 12 | NO | None | 0 | YES | LUNGS | 12 | |
| Dogan 45 | 2020 | 1 | 82 | M | C, T | G2 | TL + left ND | C. NODES (VI LEVEL) | YES | NO | NA | NR | NR | NR | NR | NR | NR | NR | |
| Adeola 46 | 2021 | 4 | NR | NR | NR | NR | NR | C. NODES, M+ | YES, 3 pts | YES, 1 pt | NR | NR | NR | NR | NR | NR | NR | NR | |
| Talati 47 | 2022 | 2 | NR | NR | NR | NR | NR | C. NODES | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | |
| Roch-Zniszczoł 48 | 2022 | 1 | 68 | M | T | G1 | Tracheotomy; TL 8 m later + RT | LIVER, LUNGS | NO | NO | DOD | 84 | NO | None | 0 | YES | LIVER, LUNGS | 84 | |
| Rüller 5 | 2022 | 2 | NR | NR | C | G2 | Laryngofissure, 3 endoscopic debulking, TL + peristomal recurrence excision + PMMF | LUNGS | NO | NO | AWD | 57.4 | NO | None | 0 | YES | LUNGS | NR | |
| NR | NR | C | G2 | 2 endoscopic excisions, 2 laryngofissures, TL | LUNGS | NO | NO | AWD | 81.3 | NO | None | 0 | YES | LUNGS | 80 | ||||
| Zeitels 49 | 2022 | 1 | 48 | M | C | G2 | PL + aortic homograft; ND + RT 1 y later | NECK, MEDIASTINUM | NO | NO | AWD | 36 | YES | C. NODES | 12 | YES | MEDIASTINUM | 12 | |
| Gazda 50 | 2024 | 2 | NR | NR | NR | G1/G2 | 1° case, NR for larynx, lung resection, pulmonary radiofrequency; 2° case, palliative care | LUNGS | NR | NR | NR | NR | NR | NR | NR | YES | LUNGS | NR | |
| Schleich 51 | 2024 | 1 | NR | NR | NR | NR | NR | M+ | NR | NR | AWD | NR | NR | NR | NR | NR | NR | NR | |
| Mirza 52 | 2025 | 1 | 77 | M | C | G1 | Intubation + biopsy | LUNGS + MEDIASTINAL AND HILAR LYMPHADENOPATHIES | NO | YES | DOD | During hospitalization | NO | None | 0 | NO | None | 0 | |
| Charous 53 | 2025 | 1 | 57 | M | C, ARY | G3/DD | Debulking; TL 2 y later; ND 5 m later | C. NODES, DERMAL AND LUNGS | NO | NO | DOD | 21 | YES | C. NODES | 5 | YES | DERMAL, LUNGS | 20 | |
| *Data reported by Nakayama et al. 31; AAW; alive and well; ARY, arytenoid; AWD, alive with disease; C, cricoid cartilage; CHT, chemotherapy; C. NODES, cervical lymph nodes; COR, corniculate cartilage; CRT, chemoradiotherapy; d, days; DD, dedifferentiated LCS; DOC, died of other causes; DOD, died of disease; E, epiglottis; F, female; FU, follow-up; G1, grade 1 LCS; G2, grade 2 LCS; G3, grade 3 LCS; m, months; M, male; M+, presence of distant metastasis; mRND, modified radical neck dissection; MY, myxoid LCS; N, number; N+, presence of regional metastasis; ND, neck dissection; NED, no evidence of disease; NR, not reported; PL, partial laryngectomy; PMMF, pectoralis major myocutaneous flap; PP, partial pharyngectomy; RFFF, radial forearm free flap; RND, radical neck dissection; RT, radiotherapy; T, thyroid cartilage; TL, total laryngectomy; TLM, transoral laser microsurgery; TT, total thyroidectomy; y, years; w, weeks. | |||||||||||||||||||
References
- Thompson L, Gannon F. Chondrosarcoma of the larynx: a clinicopathologic study of 111 cases with a review of the literature. Am J Surg Pathol. 2002;26:836-851. doi:https://doi.org/10.1097/00000478-200207000-0000
- Ferlito A, Devaney K, Mäkitie A. Differing characteristics of cartilaginous lesions of the larynx. Eur Arch Otorhinolaryngol. 2019;276:2635-2647. doi:https://doi.org/10.1007/s00405-019-05563-w
- Piazza C, Del Bon F, Grazioli P. Organ preservation surgery for low- and intermediate-grade laryngeal chondrosarcomas: analysis of 16 cases. Laryngoscope. 2014;124:907-912. doi:https://doi.org/10.1002/lary.24416
- Piazza C, Paderno A, Nicolai P. Conservative surgery for laryngeal chondrosarcoma: a review of the most recently proposed approaches. Curr Opin Otolaryngol Head Neck Surg. 2017;25:93-100. doi:https://doi.org/10.1097/MOO.0000000000000337
- Ruller K, Sittel C, Kolmel J-C. Organ preservation strategies in laryngeal chondrosarcoma. Laryngoscope. 2022;132:838-843. doi:https://doi.org/10.1002/lary.29826
- Iandelli A, Missale F, Laborai A. Surgical management and oncological outcome of non-squamous cell carcinoma of the larynx: a bicentric study. Eur Arch Otorhinolaryngol. 2022;279:299-310. doi:https://doi.org/10.1007/s00405-021-07076-x
- Piazza C, Lancini D, Zigliani G. Hemicricoidectomy with modified rotational thyro-crico-tracheal anastomosis: a newborn in the family of crico-tracheal resection and anastomosis technique. Acta Otorhinolaryngol Ital. 2023;43:382-389. doi:https://doi.org/10.14639/0392-100X-N2623
- Piazza C, Montenegro C, Tomasoni M. Aggressive subtypes of laryngeal chondrosarcoma and their clinical behavior: a systematic review. Oncol Ther. 2025;13:49-67. doi:https://doi.org/10.1007/s40487-024-00323-1
- López F, Strojan P, Ferlito A. Some considerations on the treatment and prognosis of the most common malignant tumors of the larynx. Oncol Ther. 2024;12:621-628. doi:https://doi.org/10.1007/s40487-024-00301-7
- Mahase S, Singh B, Wong R. Primary chondrosarcomas of the larynx treated with proton radiotherapy: a single institutional experience. Cancer Rep (Hoboken). 2022;5. doi:https://doi.org/10.1002/cnr2.1621
- Ferlito A, Nicolai P, Montaguti A. Chondrosarcoma of the larynx: review of the literature and report of three cases. Am J Otolaryngol. 1984;5:350-359. doi:https://doi.org/10.1016/s0196-0709(84)80005-8
- Liberati A, Altman D, Tetzlaff J. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339. doi:https://doi.org/10.1136/bmj.b2700
- Ungerecht K. Multiple chondrome und chondrosarkome des larynx und der trachea mit chondromyxosarkomatosen rezidiven. Arch Ohr Nus Kehlkopfheilk. 1951;160:158-169. doi:https://doi.org/10.1007/bf02105896
- Frenzel H. Kehlkopfchondrome und ihr charakteristisches röntgenbild [Laryngeal chondroma and its roentgenographic aspects]. Monatsschr Ohrenheilkd Laryngorhinol. 1953;87:281-286.
- Sirota H, Hurwitz A. Chondrosarcoma of the larynx. JAMA Arch Otolaryngol. 1952;56:290-293. doi:https://doi.org/10.1001/archotol.1952.00710020310007
- Bronzini E. Condrosarcoma della laringe. Ann Laringol Otol Rinol Faringol. 1959;58:497-509.
- Ghalib S, Warner E, DeGowin E. Laryngeal chondrosarcoma after thyroid irradiation. JAMA. 1969;210:1762-1763.
- Al-Saleem T, Tucker G, Peale A. Cartilaginous tumors of the larynx. Clinical-pathologic study of ten cases. Ann Otol Rhinol Laryngol. 1970;79:33-41. doi:https://doi.org/10.1177/000348947007900104
- Huizenga C, Balogh K. Cartilaginous tumors of the larynx. A clinicopathologic study of 10 new cases and a review of the literature. Cancer. 1970;26:201-210. doi:https://doi.org/10.1002/1097-0142(197007)26:1<201::aid-cncr2820260126>3.0.co;2-s
- Hyams V, Rabuzzi D. Cartilaginous tumors of the larynx. Laryngoscope. 1970;80:755-767. doi:https://doi.org/10.1288/00005537-197005000-00007
- Hellquist H, Olofsson J, Gröntoft O. Chondrosarcoma of the larynx. J Laryngol Otol. 1979;93:1037-1047. doi:https://doi.org/10.1017/s0022215100088071
- Harwood A, Krajbich J, Fornasier V. Radiotherapy of chondrosarcoma of bone. Cancer. 1980;45. doi:https://doi.org/10.1002/10970142(19800601)45:11<2769::aidcncr2820451111>3.0.co;2-x
- Escher A, Escher F, Zimmermann A. Zur klinik und pathologie chondromatöser tumoren des larynx [Clinical aspects and pathology of chondromatous tumors of the larynx]. HNO. 1984;32:269-285.
- Gray S, Blanke S, Babin R. Chondrosarcoma of the epiglottis with pulmonary metastases. Otolaryngol Head Neck Surg. Published online 1984.
- Kasanzew M, John D, Newman P. Chondrosarcoma of the epiglottis. J Laryngol Otol. 1988;102:374-377. doi:https://doi.org/10.1017/s002221510010502x
- Hakky M, Kolbusz R, Reyes C. Chondrosarcoma of the larynx. Ear Nose Throat J. 1989;68:60-62.
- Jacobs R, Stayboldt C, Harris J. Chondrosarcoma of the epiglottis with regional and distant metastasis. Laryngoscope. 1989;99:861-864. doi:https://doi.org/10.1288/00005537-198908000-00016
- Nicolai P, Ferlito A, Sasaki C. Laryngeal chondrosarcoma: incidence, pathology, biological behavior, and treatment. Ann Otol Rhinol Laryngol. 1990;99:515-523. doi:https://doi.org/10.1177/000348949009900704
- Glaubiger D, Casler J, Garrett W. Chondrosarcoma of the larynx after radiation treatment for vocal cord cancer. Cancer. 1991;68:1828-1831. doi:https://doi.org/10.1002/1097-0142(19911015)68:8<1828::aid-cncr2820680831>3.0.co;2-l
- Brandwein M, Moore S, Som P. Laryngeal chondrosarcomas: a clinicopathologic study of 11 cases, including two “dedifferentiated” chondrosarcomas. Laryngoscope. 1992;102:858-867. doi:https://doi.org/10.1288/00005537-199208000-00004
- Nakayama M, Brandenburg J, Hafez G. Dedifferentiated chondrosarcoma of the larynx with regional and distant metastases. Ann Otol Rhinol Laryngol. 1993;102:785-791. doi:https://doi.org/10.1177/000348949310201009
- Moran C, Suster S, Carter D. Laryngeal chondrosarcomas. Arch Pathol Lab Med. 1993;117:914-917.
- Fichera G, Rossi G. A rare mesenchymal tumor of the larynx. Case report and anatomo-clinical features. Pathologica. 1995;87:148-153.
- Lippert B, Claassen H, Bäumken J. Das chondrosarkom des kehlkopfes [Chondrosarcoma of the larynx]. Laryngorhinootologie. 1997;76:28-35. doi:https://doi.org/10.1055/s-2007-997382
- Jones D, Dillard S, Bradford C. Cartilaginous tumours of the larynx. J Otolaryngol. 2003;32:332-337. doi:https://doi.org/10.2310/7070.2003.11351
- Rinaggio J, Duffey D, McGuff H. Dedifferentiated chondrosarcoma of the larynx. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;97:369-375. doi:https://doi.org/10.1016/j.tripleo.2003.09.015
- Casiraghi O, Martinez-Madrigal F, Pineda-Daboin K. Chondroid tumors of the larynx: a clinicopathologic study of 19 cases, including two dedifferentiated chondrosarcomas. Ann Diagn Pathol. 2004;8:189-197. doi:https://doi.org/10.1053/j.anndiagpath.2004.04.001
- Goda J, Ferguson P, O’Sullivan B. High-risk extracranial chondrosarcoma: long-term results of surgery and radiation therapy. Cancer. 2011;117:2513-2519. doi:https://doi.org/10.1002/cncr.25806
- Böscke R, Hunold P, Noack F. Laryngeal chondrosarcoma with unusual dissemination to the humerus. ORL J Otorhinolaryngol Relat Spec. 2012;74:154-157. doi:https://doi.org/10.1159/000337822
- Domínguez-Durán E, Menoyo-Bueno A, González-Cantón J. Cutaneous metastases of laryngeal chondrosarcoma. Acta Otorrinolaringol Esp. 2014;65:373-374. doi:https://doi.org/10.1016/j.otorri.2013.02.001
- Leong S, Upile N, Lau A. Extended vertical hemilaryngectomy and reconstruction with a neovascularised tracheal autograft for advanced unilateral laryngeal tumours: a learning curve. Eur Arch Otorhinolaryngol. 2014;271:1729-1735. doi:https://doi.org/10.1007/s00405-013-2694-7
- Dubal P, Svider P, Kanumuri V. Laryngeal chondrosarcoma: a population-based analysis. Laryngoscope. 2014;124:1877-1881. doi:https://doi.org/10.1002/lary.24618
- Magliocca K, Edgar M, Corey A. Dedifferentiated chondrosarcoma of the larynx: radiological, gross, microscopic and clinical features. Ann Diagn Pathol. 2017;30:42-46. doi:https://doi.org/10.1016/j.anndiagpath.2017.05.010
- Lame C, Loum B, Diallo T. Chondrosarcoma of the thyroid cartilage: a rare laryngeal malignancy. Otorhinolaryngol Head Neck Surg. Published online 2017. doi:https://doi.org/10.15761/OHNS.1000157
- Doğan S, Vural A, Kahriman G. Non-squamous cell carcinoma diseases of the larynx: clinical and imaging findings. Braz J Otorhinolaryngol. 2020;86:468-482. doi:https://doi.org/10.1016/j.bjorl.2019.02.003
- Adeola J, Patel J, Povolotskiy R. Clinicopathologic characteristics of laryngeal chondrosarcoma: an analysis of the National Cancer Database. Auris Nasus Larynx. 2021;48:956-962. doi:https://doi.org/10.1016/j.anl.2021.02.014
- Talati V, Urban M, Patel T. Laryngeal chondrosarcoma characteristics and survival analysis in the National Cancer Database. Otolaryngol Head Neck Surg. 2022;166:101-108. doi:https://doi.org/10.1177/01945998211004578
- Roch-Zniszczoł A, Księżniak-Baran D, Chmielik E. Laryngeal chondrosarcoma treated with conventional radiotherapy – Case report and review of the literature. Pol J Pathol. 2022;73:176-179. doi:https://doi.org/10.5114/pjp.2022.119240
- Zeitels S, Baird B. Surgical treatment strategies for laryngeal chondrosarcomas: a single institution investigation. Laryngoscope. 2022;132:169-176. doi:https://doi.org/10.1002/lary.29762
- Gazda P, Baujat B, Sarini J. Functional or radical surgical treatment of laryngeal chondrosarcoma, analysis of survival and prognostic factors: a REFCOR and NetSarc-ResOs multicenter study of 74 cases. Eur J Surg Oncol. 2024;50. doi:https://doi.org/10.1016/j.ejso.2023.107315
- Schleich M, Laccourreye L, Marianowski R. Treatment strategy in laryngeal chondrosarcoma: a multicenter study of 43 cases. Eur Arch Otorhinolaryngol. 2024;281:883-890. doi:https://doi.org/10.1007/s00405-023-08248-7
- Mirza N, Veeraballi S, Rayad M. A rare case of aggressive metastatic laryngeal chondrosarcoma presenting as bilateral necrotizing pneumonia. Ear Nose Throat J. 2025;104:NP116-NP120. doi:https://doi.org/10.1177/01455613221100005
- Charous S, Yuhan B, Stenson K. Dedifferentiation of a chondrosarcoma of the larynx: a case report. Head Neck. 2025;47:E58-E63. doi:https://doi.org/10.1002/hed.28004
- Baatenburg de Jong R, van Lent S, Hogendoorn P. Chondroma and chondrosarcoma of the larynx. Curr Opin Otolaryngol Head Neck Surg. 2004;12:98-105. doi:https://doi.org/10.1097/00020840-200404000-00008
- Lopes P, Nascimento J, Rocha G. Laryngeal chondrosarcoma, a 30-year series. Acta Otorrinolaringol Esp. 2025;76:20-24. doi:https://doi.org/10.1016/j.otoeng.2024.09.004
- Chin O, Dubal P, Sheikh A. Laryngeal chondrosarcoma: a systematic review of 592 cases. Laryngoscope. 2017;127:430-439. doi:https://doi.org/10.1002/lary.26068
- Neel H, Unni K. Cartilaginous tumors of the larynx: a series of 33 patients. Otolaryngol Head Neck Surg. 1982;90:201-207. doi:https://doi.org/10.1177/019459988209000212
- Lewis J, Olsen K, Inwards C. Cartilaginous tumors of the larynx: clinicopathologic review of 47 cases. Ann Otol Rhinol Laryngol. 1997;106:94-100. doi:https://doi.org/10.1177/000348949710600202
- Lavertu P, Tucker H. Chondrosarcoma of the larynx: case report and management philosophy. Ann Otol Rhinol Laryngol. 1984;93:452-456. doi:https://doi.org/10.1177/000348948409300506
Downloads
License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright
Copyright (c) 2026 Società Italiana di Otorinolaringoiatria e chirurgia cervico facciale
How to Cite
- Abstract viewed - 683 times
- PDF downloaded - 176 times



