Head and neck
Vol. 46: Issue 1 - February 2026
The prognostic impact of perioperative metabolic variables in head and neck cancer: a prospective study
Summary
Objective. Major head and neck surgeries are associated with significant perioperative morbidity. Despite extensive metabolic parameter investigations, consistent prognostic indicators remain undefined. This study aimed to elucidate metabolic predictors for perioperative prognosis.
Methods. A prospective observational study evaluated patients undergoing major head and neck surgeries (2019-2020). Nutritional status was assessed preoperatively and on postoperative days 2 and 10 using electric bioimpedance analysis. Phase angle, a bioelectrical marker of soft-tissue mass and hydration, served as a nutritional status indicator.
Results. 37 patients were included (mean age 61.3 ± 12.5 years; 45.9% males), predominantly with oral cavity origins (75.7%). Postoperative day 10 phase angle significantly correlated with one-year survival (p = 0.014). Neutrophil-to-lymphocyte ratio (NLR) on postoperative days 2 and 10 was associated with postoperative complications (p = 0.022 and 0.026). Preoperative nutritional support demonstrated improved phase angle, optimised NLR levels, and reduced risk of complications (p = 0.023).
Conclusions. Phase angle represents a significant prognostic indicator for survival of head and neck surgical patients. Preoperative nutritional intervention shows potential in reducing complications and enhancing post-surgical metabolic outcomes.
Introduction
Major head and neck surgeries can be associated with significant perioperative complications and morbidity, related both to the surgery itself which can be complicated and long, and to patient factors such as medical background, perioperative nutritional status, and nicotine and alcohol abuse, all of which can contribute to a complicated postoperative convalescence 1-3.
While the association between perioperative nutritional or metabolic parameters and postoperative complications has been studied, the results have been inconsistent and unable to define metabolic predictors for short- or long-term prognosis, or to demonstrate improved prognosis with perioperative nutritional support in these patients 4-7. Neutrophil-to-lymphocyte ratio (NLR) is described as a factor correlated with worse prognosis among oncological patients, specifically head and neck patients 8-12.
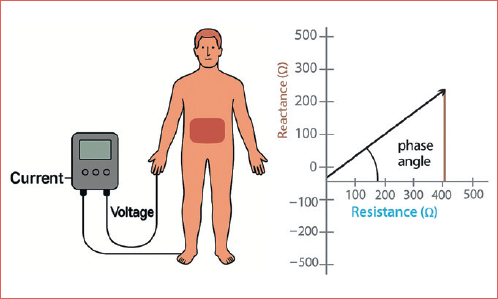
Bioelectrical impedance resistance (BIA) is a method to estimate body composition by measuring body component resistance (R) and reactance (Xc) by recording a voltage drop in applied current. It is considered a non-invasive and reliable tool to assess a patient’s nutritional status and body composition 7,8.
Phase angle, a parameter measured by BIA, indicates the ratio between resistance and reactance. It is a marker of hydration status and soft-tissue mass and considered a good indicator of the patient’s nutritional status as it represents the integrity of cell membranes 9,10. Several studies have shown that phase angle level is associated with prognosis in oncological patients 8,11-14. A study by Sat-Muñoz et al. 15 was able to demonstrate an association between phase angle and survival in head and neck cancer patients. However, no studies, to our knowledge, have demonstrated an association between phase angle level and prognosis among surgically treated head and neck cancer patients in the perioperative phase.
Our aim was to investigate the association between perioperative nutritional status – expressed by BIA factors including phase angle – and laboratory findings including NLR and postoperative complications and long term-survival and prognosis. We also attempted to describe the effect of perioperative nutritional support on survival and postoperative complications.
Materials and methods
Patients and design
We performed a prospective observation of all patients who underwent major head and neck surgery who required postoperative nasogastric tube feeding at a University-affiliated tertiary care centre between January 2019 and June 2020. All patients were followed and treated by our multi-disciplinary head and neck team, including several head and neck surgeons, head and neck oncologist, and a professional dietitian who assessed patients preoperatively and prescribed an oral supplement of immune-modulating nutrition (Impact, Nestle, Switzerland) to all patients in the perioperative period (5 days before surgery and during the first 5 postoperative days). Patients who experienced delirium and mental status change postoperatively were excluded from the cohort due to technical difficulty in recording their BIA parameters. All patients enrolled in the study gave their informed consent prior to enrollment. Data were collected from patients’ medical charts and included demographics, risk factors, clinical features, staging, treatment modalities, postoperative complications and outcomes. Patients’ nutritional status was assessed at 3 time points – one day before surgery, and at 2 and 10 days after surgery. At these time points, height and weight were measured for body mass index (BMI) calculation, blood analysis was obtained including complete blood count, serum albumin, pre-albumin, transferrin, iron and C-reactive protein (CRP), and erythrocyte sedimentation rate (ESR). NLR was calculated by dividing the absolute number of neutrophils by the absolute value of lymphocytes. As the median value of NLR for all patients on the preoperative evaluation was 5.1, we decided to use a cut-off of 5 as the ratio to define NLR as high or low. Body composition measures were performed using the Quadscan 4000 (Bodystat®, UK) multi-frequential electric BIA apparatus. Based on a previous study from our institute 9, we used a cut-off of 5° to define the level of phase angle as low or high. The Cover figure illustrates the methodology of BIA and phase angle measurement as applied in our perioperative nutritional assessment protocol.
Statistical analysis
Differences in mean continuous variables between groups were analysed by Student’s T-test and Mann-Whitney test. Differences in categorical variables were determined by Fisher’s exact test. Multi-variate analysis was performed using the Cox regression test. Rates of disease-specific, disease-free, and overall survival were calculated from the date of diagnosis to the date of death or recurrence or last follow-up. These data were analysed by the Kaplan-Meier method and log-rank test was used to determine significance. Analyses were conducted using IBM SPSS Statistics for Windows, Version 26.0. (IBM Corp. Armonk, NY).
The study was approved by the medical centre ethics board. (Institutional Review Board approval number RMC-18-255) and informed consent was waived.
Results
Patients
A total of 37 patients were included in the study. Three additional patients were initially enrolled but excluded due to postoperative delirium. Data regarding tumour origin, staging, and the surgical procedure performed is presented in Table I. The mean age of all patients was 61.3 + 12.5 years and 17 were males (45.9%). Ten patients (27%) were smokers and 8 were past smokers (21.6%). Four patients were regular alcohol consumers (10.8%). While all patients were recommended perioperative nutrition support after being evaluated by a dietitian, 29 patients (78.4%) eventually received it. Most tumours originated in the oral cavity (75.7%), and all patients underwent ipsilateral neck dissection, either elective or therapeutic, while 10 (27%) had bilateral neck dissection. Reconstruction was performed in 28 cases (75.7%), with a free flap used in 17 cases (46%).
Prognostic role of laboratory factors
Data regarding metabolic and laboratory variables is presented in Figure 1. When analysing laboratory data, including complete blood count, NLR, ESR, CRP, albumin and pre-albumin, we were able to demonstrate a significant increase in NLR on postoperative day (POD) 2 compared to its level at one day before surgery and on POD 10. NLR level above 5 on POD 2 was significantly associated with disease-specific survival (log rank p value = 0.021, Fig. 2) and marginally associated with overall survival (log rank p value = 0.056, Fig. 3). NLR levels one day prior to the surgery and on POD 10 were not associated with survival. However, NLR level above 5 on POD 10 was associated with postoperative complications (75% vs 33.3%, p = 0.012). Postoperative complications were significantly associated with one year survival (p = 0.024). A significant increase in ESR levels was also demonstrated in POD 2 and POD 10. A significant decrease in pre-albumin level was shown on POD 2 compared to both the preoperative day and to POD 10.
Prognostic role of bioimpedance factors
When analysing changes in phase angle values, we found a significant decrease in POD 2 relative to the preoperative day and POD 10 (6.43° on the preoperative day, 4.85° on POD 2 and 5.97° on POD 10, p value = 0.002, Fig. 1). Phase angle level lower than 5° on POD 10 was significantly associated with worse disease-specific survival (Log rank p value = 0.039, Fig. 4) and overall survival (Log rank p value = 0.02, Fig. 5). On multivariate analysis that included several demographic, laboratory, and metabolic factors, a trend toward significance was noted regarding the association between low phase angle on POD 10 and survival (Odds Ratio – 8.9, p = 0.09, Tab. II). Low phase angle level on POD 2 was significantly associated with postoperative complications (40.5% vs 10.8%, p = 0.005) and a trend toward significance was also noted for the association between low level on POD 10 and postoperative complications (66.7% vs 36.8%, p = 0.07). Phase angle level one day prior to the operation was not associated with survival or complications.
Prognostic role of nutritional factors
Patients who received preoperative nutritional support (n = 29) – including the addition of caloric and protein formula during the 5 days prior to surgery and the 5 postoperative days – had a significantly lower rate of postoperative complications (41.2%, vs 87.5%, p = 0.023). Furthermore, preoperative nutritional support was also associated with better phase angle profile one day prior to surgery (p = 0.038), and better NLR profile at POD 2 (p = 0.052) and POD 10 (p < 0.001). No difference regarding mean weight or BMI was demonstrated between preoperative day and POD 2 and POD 10.
Discussion
Our results demonstrate that low postoperative phase angle on POD 10 is associated with worse overall and disease-specific survival, and that higher postoperative NLR on POD 2 is associated with worse disease-specific survival and postoperative complications. Furthermore, perioperative nutritional support may reduce postoperative complications rate and is associated with better perioperative phase angle and NLR profiles.
Patients with head and neck cancer are especially prone to malnutrition and cancer cachexia, with around 60% of patients presenting with malnutrition at diagnosis 16. Possible explanations for this high rate of malnutrition include mechanical obstruction, dysphagia, and catabolic state secondary to the tumour itself 3,9,17,18. Malnutrition among head and neck cancer patients is considered as an independent negative prognostic factor 5,19.
NLR was significantly associated with postoperative complications in our cohort, both at POD 2 and at POD 10. These results intensify the prognostic role of NLR as a predictor of outcome among head and neck cancer patients, as demonstrated by a meta-analysis published by Mascarella et al. 12.
The importance of phase angle as a prognostic factor among head and neck cancer patients was described in a publication by Sat-Muñoz et al. 21. In their cohort, patients with head and neck malignancy with low phase angle (n = 74) had a significantly lower survival rate compared to patients with normal phase angle (n = 65). Their cohort, however, did not include surgical patients and did not attempt to study the prognostic significance of perioperative phase angle level, as was performed in our study. Gupta et al. 18 compared 129 breast cancer patients with a phase angle lower than or equal to 5.6°, with 130 similar patients with phase angle above 5.6°. They were able to demonstrate a significant difference in survival, as the median survival of the first group was 23.1 months compared with 49.9 months for the second group (p = 0.021). In our cohort, we were able to demonstrate a significant association between POD 10 phase angle level and survival. Patients whose phase angle was above 5° had a significantly better overall and disease-specific survival rate compared to those with a phase angle lower than 5° (log-rank p value = 0.02 and log-rank p value = 0.039, respectively). These results demonstrate that the metabolic and nutritional status of the patient – as reflected by phase angle – during the perioperative period carries a prognostic significance and should be addressed in every head and neck cancer patient, especially during the pre- and postoperative period. While multivariate analysis of factors associated with survival on POD 10 demonstrated only a marginally significant association for phase angle level on POD 10 (p = 0.09), we believe that this result is still meaningful and demonstrates that phase angle is a significant marker that reflects the metabolic status of the head and neck patient in the perioperative phase, enabling the multidisciplinary team to identify patients whose postoperative metabolic status requires further follow-up and support. We believe that with a larger cohort, the prognostic effect of phase angle could also be significant on multivariate analysis. Low postoperative phase angle level may also help identify patients who do not seem to meet their nutritional requirements. This issue is particularly important and relevant among head and neck cancer patients, as feeding may be disturbed possibly due to pain, trismus, or mechanical obstruction, as mentioned above. These patients may be candidates for a surgical feeding solution, either temporary or constant. As we demonstrated that the postoperative metabolic state is significantly associated with overall prognosis, the need for a surgical feeding solution should be recognised and met in an early stage – during the perioperative period and not postponed for a later time.
Other studies also attempted to define the role of phase angle in the perioperative period. A study by Zhou et al. 14 demonstrated a significant association between preoperative phase angle level and postoperative complication rate among patients with pancreatic cancer undergoing pancreatico-duodenectomy. Yu et al. 20 showed an association between low phase angle level and postoperative complication rate among gastric cancer patients undergoing gastrectomy. Our results demonstrate – for the first time in head and neck cancer patients – that postoperative phase angle is specifically correlated with postoperative complications and survival. However, the level of phase angle one day prior to the operation was not associated with prognosis in our cohort, meaning that the significance of this association may be more relevant for prognosis in the postoperative setting than at any point after the diagnosis of the malignancy prior to surgery.
A significant decrease in phase angle level during radiotherapy treatment for head and neck cancer was described by Kohli et al. 19, also reflecting the gradual deterioration in nutritional and metabolic status among these patients during treatment. As many patients who undergo major head and neck surgeries require adjuvant radiotherapy, these results are relevant and further emphasise the importance of continuous monitoring of nutritional and metabolic status during the surgical and oncological treatment phases, and proper nutritional support as indicated.
We were able to demonstrate that perioperative nutritional support improved the NLR and phase angle profiles of our cohort and reduced postoperative complications. These results are a further testimony to the important role of diagnosing and treating malnutrition among head and neck patients prior to surgical treatment 20. In order to achieve this goal, the multidisciplinary team must be aware of the high prevalence of malnutrition in these patients, and to the fact that patients with head and neck cancer and malnutrition are prone to worse survival and more postoperative complications. BIA offers an easy and non-invasive way to evaluate the nutritional and metabolic status of the patient, and NLR provides another easy and inexpensive assessment tool for postoperative status and prognosis. Awareness of the management team to these tools can aid in early diagnosis of malnutrition, and in turn proper treatment by a nutritionist, surgical feeding solutions when necessary, and close follow-up of the patient’s metabolic status by both the surgeon and oncologist during the entire treatment process.
The limitations of our study include mainly the relatively small number of participants. In addition, we did not use any measurement tool to assess nutritional status other than laboratory exams and BIA. However, since BIA factors were already demonstrated to correlate with nutritional status in several studies 7,8, we felt comfortable using these assessment tools.
Conclusions
BIA and phase angle specifically enables non-invasive assessment of postoperative metabolic status among head and neck cancer patients. The postoperative level of phase angle and NLR are associated with overall survival and postoperative complications. Monitoring these factors in the postoperative phase may assist the treatment team in early identification and treatment of malnutrition among these patients, and consequently reduce the rate of perioperative complications and improve overall survival.
Conflict of interest statement
The authors declare no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author contributions
EY: substantial contributions to conception and design of the study, data analysis, drafting of manuscript, final approval and accountability for the work;,YS: data acquisition, drafting of manuscript, final approval and accountability for the work; OZ: data analysis, final approval and accountability for the work; NK: data analysis, substantial contributions to conception and design of the study, final approval, and accountability for the work; PS: data analysis and interpretation, drafting of manuscript, final approval, and accountability for the work; MY: data analysis and interpretation, final approval, and accountability for the work; TS: data interpretation, drafting of manuscript, final approval and accountability for the work; AM: substantial contributions to conception and design of the study, drafting of manuscript, final approval and accountability for the work; GB: substantial contributions to conception and design of the study, drafting of manuscript, final approval, and accountability for the work; ST: substantial contributions to conception and design of the study, data analysis, drafting of manuscript, final approval and accountability for the work.
Ethical consideration
This study was approved by the Institutional Ethics Committee (Rabin Medical Center [Institutional Review Board approval number RMC-18-255]).
The research was conducted ethically, with all study procedures being performed in accordance with the requirements of the World Medical Association’s Declaration of Helsinki.
Written informed consent was obtained from each patient for study participation and data publication.
History
Received: November 26, 2024
Accepted: July 7, 2025
Figures and tables
Figure 1. Changes in metabolic and laboratory factors levels according to the time of surgery.
Figure 2. Kaplan-Meier analysis comparing disease-specific survival between patients with NLR ratio above and below 5 on POD 2.
Figure 3. Kaplan-Meier analysis comparing overall survival between patients with NLR ratio above and below 5 on POD 2.
Figure 4. Kaplan-Meier analysis comparing disease-specific survival between patients with phase angle level above and below 5° on POD 10.
Figure 5. Kaplan-Meier analysis comparing overall survival between patients with phase angle level above and below 5° on POD 10.
| Variable | n = 37 |
|---|---|
| (%) | |
| Gender | |
| Female | 20 (54.1) |
| Male | 17 (45.9) |
| Age, years | |
| 61.3 ± 12.5 | |
| Smoking | |
| Active smoker | 10(27) |
| Past smoker | 8 (21.6) |
| No | 19 (51.4) |
| Alcohol | |
| Yes | 4 (10.8) |
| No | 33 (89.2) |
| Origin site | |
| Tongue | 13 (35.1) |
| Alveolar ridge | 5 (13.5) |
| Floor of mouth | 4 (10.8) |
| Buccal mucosa | 2 (5.4) |
| RMT | 2 (5.4) |
| Hard palate | 2 (5.4) |
| Larynx | 4 (10.8) |
| Base of tongue | 1 (2.7) |
| Tonsil | 1 (2.7) |
| Thyroid | 1 (2.7) |
| Maxilla | 1 (2.7) |
| Submandibular (UP) | 1 (2.7) |
| Surgery | |
| Excision of oral cavity tumour | 28 (75.7) |
| Total laryngectomy | 5 (13.5) |
| Total laryngo-pharyngectomy | 1 (2.7) |
| Crico-tracheal resection | 1 (2.7) |
| Maxillectomy | 1 (2.7) |
| TORS + cervical approach | 1 (2.7) |
| Neck dissection | |
| Ipsilateral | 27(73) |
| Bilateral | 10(27) |
| Reconstruction | |
| Yes | 28 (75.7) |
| Local flap | 4 (10.8) |
| Regional flap | 6 (16.2) |
| Free flap | 17(46) |
| Gastric pull-up | 1 (2.7) |
| No | 9 (24.3) |
| Tracheostomy during surgery | |
| Yes | 26 (70.3) |
| No | 11 (29.7) |
| Perioperative nutritional support | |
| 29 (78.4) | |
| Gastrostomy during surgery | |
| Yes | 4 (10.8) |
| No | 33 (89.2) |
| Postoperative complications | |
| 19 (51.4) | |
| Pathologic T classification | |
| T1 | 2 (5.4) |
| T2 | 9 (24.3) |
| T3 | 5 (13.5) |
| T4 | 15 (40.5) |
| No data | 6 (16.2) |
| Pathologic N classification | |
| N0 | 15 (40.5) |
| N1 | 4 (10.8) |
| N2 | 8 (21.6) |
| N3 | 5 (13.5) |
| No data | 5 (13.5) |
| Distant metastasis | |
| Yes | 0 |
| No | 37(100) |
| RMT: retro-molar trigone; UP: unknown primary; TORS: transoral robotic surgery. | |
| Variable | Multivariate analysis | Multivariate analysis | Lower CI | Upper CI |
|---|---|---|---|---|
| p value | Odds ratio | |||
| Age | 0.79 | 1.013 | 0.922 | 1.112 |
| Gender | 0.89 | 0.87 | 0.13 | 5.88 |
| Phase angle below 5° at POD 10 | 0.09 | 8.87 | 0.67 | 117.35 |
| BMI above 25 at POD 10 | 0.58 | 0.47 | 0.033 | 6.82 |
| ESR above 30 at POD 10 | 0.39 | 0.34 | 0.029 | 4 |
| PLT above 450 at POD 10 | 0.27 | 0.27 | 0.026 | 2.81 |
| Pre albumin below 15 at POD 10 | 0.1 | 9.42 | 0.63 | 141.2 |
| Advanced stage | 0.07 | 0.062 | 0.003 | 1.28 |
| POD: postoperative day; BMI: body mass index; ESR: erythrocyte sedimentation rate; PLT: platelets; CI: confidence interval. | ||||
References
- Parhar H, Durham J, Anderson D. The association between the Nutrition-Related index and morbidity following head and neck microsurgery. Laryngoscope. 2020;130:375-380. doi:https://doi.org/10.1002/lary.27912
- Gourin C, Couch M, Johnson J. Effect of weight loss on short-term outcomes and costs of care after head and neck cancer surgery. Ann Otol Rhinol Laryngol. 2014;123:101-110. doi:https://doi.org/10.1177/0003489414523564
- Chasen M, Bhargava R. A descriptive review of the factors contributing to nutritional compromise in patients with head and neck cancer. Support Care Cancer. 2009;17:1345-1351. doi:https://doi.org/10.1007/s00520-009-0684-5
- van Bokhorst-De Van Der Schueren M, Quak J, von Blomberg-van der Flier B. Effect of perioperative nutrition, with and without arginine supplementation, on nutritional status, immune function, postoperative morbidity, and survival in severely malnourished head and neck cancer patients. Am J Clin Nutr. 2001;73:323-332. doi:https://doi.org/10.1093/ajcn/73.2.323
- Cantwell L, Fahy E, Walters E. Nutritional prehabilitation in head and neck cancer: a systematic review. Support Care Cancer. 2022;30:8831-8843. doi:https://doi.org/10.1007/s00520-022-07239-4
- Tomasoni M, Piazza C, Deganello A. The prognostic-nutritional index in HPV-negative head and neck squamous cell carcinoma treated with upfront surgery: a multi-institutional series. Acta Otorhinolaryngol Ital. 2023;43:170-182. doi:https://doi.org/10.14639/0392-100X-N2358
- Boscolo-Rizzo P, Zanelli E, Giudici F. Prognostic value of H-index in patients surgically treated for squamous cell carcinoma of the larynx. Laryngoscope Investig Otolaryngol. 2021;6:729-737. doi:https://doi.org/10.1002/lio2.603
- Mattavelli D, Lombardi D, Missale F. Prognostic nomograms in oral squamous cell carcinoma: the negative impact of low neutrophil to lymphocyte ratio. Front Oncol. 2019;9. doi:https://doi.org/10.3389/fonc.2019.00339
- Marchi F, Missale F, Incandela F. Prognostic significance of peripheral T-cell subsets in laryngeal squamous cell carcinoma. Laryngoscope Investig Otolaryngol. 2019;4:513-519. doi:https://doi.org/10.1002/lio2.304
- Gaudioso P, Borsetto D, Polesel J. Blood markers predicting clinically occult lymph node metastasis in head and neck squamous cell carcinoma. ORL J Otorhinolaryngol Relat Spec. 2024;86:32-40. doi:https://doi.org/10.1159/000534079
- Mazzocco T, Migliorelli A, Tirelli G. Prognostic value of changes in pre- and postoperative inflammatory blood markers in HPV-negative head and neck squamous cell carcinomas. Acta Otorhinolaryngol Ital. 2025;45:10-20. doi:https://doi.org/10.14639/0392-100X-N2955
- Mascarella M, Mannard E, Silva S. Neutrophil-to-lymphocyte ratio in head and neck cancer prognosis: a systematic review and meta-analysis. Head Neck. 2018;40:1091-1100. doi:https://doi.org/10.1002/hed.25075
- Zhang X, Zhao W, Du Y. A simple assessment model based on phase angle for malnutrition and prognosis in hospitalized cancer patients. Clin Nutr. 2022;41:1320-1327. doi:https://doi.org/10.1016/j.clnu.2022.04.018
- Zhou S, Yu Z, Shi X. The relationship between phase angle, nutrition status, and complications in patients with pancreatic head cancer. Int J Environ Res Public Health. 2022;19. doi:https://doi.org/10.3390/ijerph19116426
- Tzelnick S, Singer P, Shopen Y. Bioelectrical impedance analysis in patients undergoing major head and neck surgery: a prospective observational pilot study. J Clin Med. 2021;10. doi:https://doi.org/10.3390/jcm10030539
- Barbosa-Silva M, Barros A, Wang J. Bioelectrical impedance analysis: population reference values for phase angle by age and sex. Am J Clin Nutr. 2005;82:49-52. doi:https://doi.org/10.1093/ajcn.82.1.49
- Morlino D, Cioffi I, Marra M. Bioelectrical phase angle in patients with breast cancer: a systematic review. Cancers (Basel). 2022;14. doi:https://doi.org/10.3390/cancers14082002
- Gupta D, Lammersfeld C, Vashi P. Bioelectrical impedance phase angle as a prognostic indicator in breast cancer. BMC Cancer. 2008;8. doi:https://doi.org/10.1186/1471-2407-8-249
- Kohli K, Corns R, Vinnakota K. A bioimpedance analysis of head-and-neck cancer patients undergoing radiotherapy. Curr Oncol. 2018;25:E193-E199. doi:https://doi.org/10.3747/co.25.3920
- Yu B, Park K, Park J. Bioelectrical impedance analysis for prediction of early complications after gastrectomy in elderly patients with gastric cancer: the phase angle measured using bioelectrical impedance analysis. J Gastric Cancer. 2019;19:278-289. doi:https://doi.org/10.5230/jgc.2019.19.e22
- Sat-Muñoz D, Martínez-Herrera B, González-Rodríguez J. Phase angle, a cornerstone of outcome in head and neck cancer. Nutrients. 2022;14. doi:https://doi.org/10.3390/nu14153030
- Jager-Wittenaar H, Dijkstra P, Dijkstra G. High prevalence of cachexia in newly diagnosed head and neck cancer patients: an exploratory study. Nutrition. 2017;35:114-118. doi:https://doi.org/10.1016/j.nut.2016.11.008
- George J, Cannon T, Lai V. Cancer cachexia syndrome in head and neck cancer patients: part II. Pathophysiology. Head Neck. 2007;29:497-507. doi:https://doi.org/10.1002/hed.20630
- Couch M, Lai V, Cannon T. Cancer cachexia syndrome in head and neck cancer patients: part I. Diagnosis, impact on quality of life and survival, and treatment. Head Neck. 2007;29:401-411. doi:https://doi.org/10.1002/hed.20447
- Correia M, Waitzberg D. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:https://doi.org/10.1016/s0261-5614(02)00215-7
- Langius J, Zandbergen M, Eerenstein S. Effect of nutritional interventions on nutritional status, quality of life and mortality in patients with head and neck cancer receiving (chemo)radiotherapy: a systematic review. Clin Nutr. 2013;32:671-678. doi:https://doi.org/10.1016/j.clnu.2013.06.012
Downloads
License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright
Copyright (c) 2026 Società Italiana di Otorinolaringoiatria e chirurgia cervico facciale
How to Cite
- Abstract viewed - 415 times
- PDF downloaded - 140 times



