Head and neck
Vol. 46: Issue 1 - February 2026
Beyond sentinel node biopsy: preliminary results of indocyanine green-guided neck dissection in cN0 tongue cancer
Summary
Objective. Neck management in oral cancer is crucial, as regional involvement strongly affects survival. Elective dissection leaves out 10% of recurrences, mainly at levels IV-V or contralateral. Sentinel node biopsy is a different strategy to detect unexpected lymphatic drainage. Our aim is to develop a tumour-specific lymphatic mapping approach for locally-advanced cN0 tongue cancers by combining scintigraphy biopsy with indocyanine green (ICG) guidance.
Methods. In this multicentre prospective pilot study lymphatic drainage was evaluated preoperatively using scintigraphy and intraoperatively with ICG fluorescence. Bilateral neck dissection was first performed removing ICG enhancing nodes, then those identified by gamma probe and lastly the remaining ones.
Results. Nine patients were enrolled. Robbin’s levels mostly enhanced were IB-IIA-III (83%), followed by level IV (66%), IIB (50%) and IA (33%). The ICG method showed a sensitivity of 100%, a positive predictive value of 47% compared to lymphoscintigraphy and of 6.3% compared to metastatic nodes. A negative predictive value of 100% was observed by both.
Conclusions. A tailored lymphography-guided neck dissection could provide an effective lymphatic mapping with any individual variation allowing to overcome the limitations of sentinel biopsy and pitfalls associated with traditional elective treatment.
Introduction
Cervical node involvement is one of the most important prognostic factors in oral tongue cancer. The presence of even a single metastatic localisation has been associated with a reduction in overall survival of up to 50% 1. The management of cervical nodes is thus as crucial as the treatment of the primary site. Nevertheless, this issue remains a subject of debate, especially regarding the management of cN0 cases.
Several studies have shown that the gold standard elective neck dissection (END) detects occult neck metastases in only approximately 20-30% of cN0 patients, leaving nearly two-thirds of patients undergoing unnecessary ND along with its associated morbidity and surgical risks 2,3.
On the other hand, a conservative “wait and see” strategy (i.e. treating only those who develop nodal metastasis during postoperative follow-up) has shown poorer survival outcomes 4.
Since the early 19th century, the concept and surgical techniques of ND have evolved significantly. From the upfront radical approach ND has gradually transitioned towards a more conservative one, aiming to preserve functional structures without compromising oncological results 5.
Most authors generally recommend a selective ND of levels I to III 6. This recommendation is purely based on statistical analyses of the risk of nodal involvement at each specific neck level. However, the lymphatic pathways of the oral cavity exhibit high complexity and variability. Some authors argue on the inclusion of level IV, V, or IIB 7,8. Additionally there is a noteworthy concern regarding the risk of contralateral occult metastases which may be missed by ipsilateral selective ND, particularly when native lymphatic drainage is bilateral. Recurrence has been reported in 10% of elective ND cases, mainly at levels IV, V and in the contralateral neck 6,9. These so-called ‘skip metastases’ exemplify the variability of the oral cavity’s lymphatic drainage, which may not be entirely included in a standardised I-III ND.
A different strategy for nodal management is represented by sentinel lymph node biopsy (SLB), which consists in sampling the first lymph node to receive drainage from primary tumour’s lymphatic stream 10.
The SLB technique made a revolution in the treatment of early breast cancer, improving breast cancer staging and treatment and avoiding a considerable amount of elective axillary lymph node dissection 11.
Neck lymphatic drainage from the oral cavity is significantly more complex than that of breast cancer, making the application of SLB more challenging. Despite numerous studies in its early years, the efficacy of SLB has thus produced less promising results 12,13. In this scenario, De Cicco et al. recommended the use of preoperative lymphoscintigraphy to create a comprehensive lymphatic map of the tumour region in a selected cohort of patients with T2-4 squamous cell carcinoma of the tongue. Their goal was to test the hypothesis that cervical node removal based on lymphatic mapping could provide a thorough approach for managing the neck in cN0 cases 14.
A significant advancement in lymph node localisation for oral cancer occurred with the integration of fluorescence imaging with indocyanine green (ICG) 15. This technique allows a direct intraoperative detection of the lymphatic drainage of the mucosal area infiltrated by the tumour along with the T-N tract 16.
A recent review on the role of SLB reports the growing of this technique supported by multicentre studies and meta-analysis 20. Due to the high identification rate, the low false-negative rate and its reasonable outcomes, the National Comprehensive Cancer Network (NCCN) recognises the use of SLB as an alternative to elective neck dissection in oral cancer. Conversely, current American Society of Clinical Oncology (ASCO) guidelines do not provide any specific recommendations. To date, this approach serves primarily as a diagnostic procedure, enabling the avoidance of END in cases with negative results. However, its main limitation is the 10-15% false negative rate, which may require a second surgery to complete the neck dissection in a previously operated field 21.
Our study introduces a novel approach to lymph node treatment in tongue cancer, aiming to improve nodal management by identifying and removing not only a single lymph node but the entire lymphatic network involved, as detected by intraoperative visualisation of the lymphatic drainage from the infiltrated tongue area.
The present pilot study aims to leverage the experience gained with lymphatic drainage studies through ICG and to evaluate its application as an intraoperative method to visualise the real lymphatic pathway from tongue cancer, whether it is ipsilateral or contralateral. Moreover, we aim to assess a tailored ND by removing a limited number of lymph nodes involved by the pathological lingual drainage area in a short series of locally advanced cN0 tongue cancers.
Materials and methods
This multicentre prospective pilot study was performed at the Otolaryngology Departments of the European Institute of Oncology and Hospital of Bolzano. The inclusion criteria were: age ≥ 18 years, a primary tumour involving the lateral border of the mobile tongue without any midline involvement, histologically confirmed diagnosis of squamous cell carcinoma, clinically staged cT4 (either due to bone/skin invasion or size/depth of invasion, according to the 8th edition of the TNM classification) in absence of lymph node (cN0) and distant metastases (cM0).
The study was proposed to 20 patients meeting the inclusion criteria at the European Institute of Oncology in Milan for oral cancer. Among them, 8 patients agreed to participate and were enrolled, with only 6 cases ultimately analysed. Two patients were excluded: one due to discrepancies between reported lymph node levels during surgery and those found in the pathological anatomy report, and another who requested exclusion from the final study. Similarly, of the 6 cases proposed from the Hospital of Bolzano, only 3 patients met the inclusion criteria and agreed to participate. Overall, 9 patients between the 2 centres were submitted to the study.
All patients were clinically and radiologically staged with a magnetic resonance imaging (MRI) with contrast enhancement and positron emission tomography (PET)/computed tomography (CT). Patient’s lymphatic drainage was studied both preoperatively with radiocolloid technetium 99 (99mTc) lymphoscintigraphy (LS) and intraoperatively with the ICG fluorescence.
The traditional radiocolloid lymph node study was thus integrated intraoperatively with the innovative fluorescent imaging method, through perilesional injection of the submucosal tongue with ICG and the use of a special near infrared (NIR) fluorescence camera (Photodynamic Eye Camera, PDE) (Cover figure).
Additionally, the lymphatic connection pathways between the primary tumour and cervical nodes (T-N tract) were analysed in detail.
All patients underwent major surgery with a combined approach, flap reconstruction and bilateral ND, which was first performed removing the lymphatic tracts identified with the ICG, then the residual ones detected by the gamma probe. Finally, ND was completed removing the remaining non-enhancing nodes, as the traditional END require.
Study design
Pre-surgical steps:
- The day prior to surgery, a planar lymphoscintigraphy (dynamic and static) marking the skin projection of the enhancing lymph nodes of the neck was performed.
Surgical steps
- Peritumoural ICG injection and identification of the lymphatic network highlighted by the fluorescent tracer through a PDE.
- Identification and removal of cervical nodes and lymphatic tracts (ipsilateral and contralateral) identified by ICG fluorescence.
- Investigation of the removed fluorescent lymph node using a gamma probe, to determine which and how many were radio-positive too (hot).
- After excising the nodes identified through ICG imaging, a comprehensive inspection of the neck was performed using a gamma probe to detect any additional radio-positive lymph nodes that might not have been fluorescent under ICG visualisation (white).
- Completion of functional ND bilaterally according to the study protocol (I-V) to thoroughly assess lymphatic drainage across all possible variations.
- Removal of the primary tumour en bloc along with the T-N tract and the residual neck dissection with a pull-through technique or by means of mandibulotomy.
- Surgical specimen’s inspection and lymph nodes/lymphatic pathway marking for histopathological examination. Anatomic localisation of sentinel lymph nodes (SLNs) was reported according to Robbins’ classification of nodal levels.
- Macro-sections’ preparation in an anatomic laboratory containing the entire lymphatic connection between the primary tumour (T) and the cervical lymph nodes (T-N tract).
- Histological examination of the entire specimen.
Radiocolloid lymphoscintigraphy method
All patients underwent planar, dynamic and static lymphoscintigraphy the day prior to surgery or 2 hours prior to the procedure.
We used colloidal particles of human albumin with dimensions between 20 and 80 nanometers (Nanocoll), labelled with 99mTc and filtred to obtain only molecules of the smallest diameter (< 50 nm), to favour their drainage and diffusion through neck interstitial tissues. Approximately 7-10 mBq of the radioactive tracer was prepared in a solution of 0.2 ml+0.2 ml of air volume and inoculated with a 25 G needle. The injection was performed into the submucosal/peritumoral area throughout 3 points (7-10 MBqx3).
Pre-surgical dynamic and static scintigraphic images were collected using a single-head gamma camera (Starcam 4000 GE Medical System, Milwaukee, WI, USA). Tomographic images were obtained using a dual-head gamma camera (Millennium GE Medical System, Milwaukee, WI, USA). Both gamma cameras were equipped with a low energy collimator with high resolution.
Patients were placed in supine position with the head approximately in the same position as that of the surgery. Dynamic acquisitions started immediately after radiocolloid administration and continued for 15 minutes (30 frames/sec), while static images, in anteroposterior and lateral projections, were acquired 30 minutes and 2 hours following the injections. At the end of the lymphoscintigraphic procedure, the skin projection of the underlying enhancing lymph nodes was marked with India ink. Scintigrafic data were matched to CT images in order to achieve a topografic localisation of the interested lymphatic drainage on both coronal and axial CT scans (Fig. 1).
During the surgical procedure, the exact location of radioactive node was further verified using a gamma probe (C-Trak System, Care-Wise, CA, USA), as in radioguided surgery (RGS). At the end of procedure, an overall check of the entire surgical field was performed in order to detect any residual radioactivity. Lastly, the surgical specimen was firstly analysed with a gamma camera to properly match radioactive areas to lymph nodes, and then histologically evaluated.
Indocyanine green fluorescence method
Following subplatismal flap harvesting, we intraoperatively performed 3 to 4 submucosal/peritumoral injections of 1-2 ml at 5 mg/ml of ICG (Fig. 2). ICG (ICG-Pulsion; Pulsion Medical Systems) has been previously prepared as powder suspension into sterile water for injectable solutions at a weight/volume ratio of 5 mg/ml.
Once injected, the ICG allowed to obtain NIR reflectance fluorescence imaging through the use of PDE (Neo, Hamamatsu Photonics, Hamamatsu, Japan) or directly on a monitor (Exoscope Storz OPAL1, Karl Storz), and to follow the transcutaneous course of the tumoural lymphatic drainage (up to 3 cm of depth).
Once the ICG has reached cervical lymphatic pathways (approximately 15-20 min later), the visual signal given by the fluorescence enables surgeons to identify the entire lymphatic network along with the enhancing and potentially pathological nodes. Similarly to the RGS, a final check of any residual fluorescence confirmed the sentinel lymph node removal.
Criteria for identifying enhancing lymph nodes:
- in case of agreement between the accumulation area of the radiopharmaceutical and fluorescent tracer, the SLNs were identified as enhancing LN;
- in instances of discrepancy, SLNs labelled with the radioisotope were designated as radio-enhanced lymph nodes (hot), while those labelled with ICG (green) were categorised as other lymphatic tissue.
All the removed lymph nodes were sent for histopathological examination, and the outcome of each specimen analysed was recorded. Each node was therefore classified according to fluorescence ICG positivity or negativity, radiocolloid 99mTc positivity or negativity and absence or presence of neoplastic cells.
The data collection aimed to extrapolate and define the percentage of agreement between the 2 methods, thereby determining the identification rate and sensitivity of fluorescence lymphography in detecting the tumour’s lymphatic network, in comparison to the results of radiocolloid lymphoscintigraphy and anatomopathological analysis of the specimen.
No risks related to the procedures were highlighted. Fluorescence imaging does not interfere with and does not alter the results and reliability of the radiocolloid method (current gold standard). The 99mTc lymphoscintigraphic procedure and the ICG fluorescence imaging method were both well tolerated by all patients, with no evidence of side effects.
Statistical analysis
This is a method-validating pilot study, which can be classified among feasibility studies. The number of patients, therefore, is not based on statistical hypotheses to be tested. The objective is mainly observational, aiming to assess the consistency of the results obtained from the 2 methods. Hence, we evaluated the percentage of agreement between lymph nodes identified using ICG lymphography (green LN) and those detected by 99mTc-radiocolloid lymphoscintigraphy (hot LN).
The possible combinations derived from the two methods are shown in Table I, considering the radioactive tracer (± vital ICG) as gold standard. The key quantities of interest in our study are as follows: A (true positives), B (false positives), and C (false negatives).
We calculated the sensitivity of the innovative ICG method compared to traditional lymphoscintigraphy as the percentage of lymph nodes identified both through ICG and the gold standard (A), relative to the total number of lymph nodes identified solely by the gold standard (A + C). Furthermore, the positive predictive value (PPV) was calculated as the percentage of LNs identified both through ICG and the gold standard (A), out of the total LNs identified through ICG (A + B). Negative predictive value (NPV) was calculated as the percentage of LNs negative to the gold standard and ICG (D), out of the total LNs solely negative to ICG (C + D). Subsequently, we calculated PPV and NPV by evaluating the ability of the innovative method to identify lymph nodes with metastatic cells.
Results
Data on patients who participated in the study (8 males and one female; mean age 56 years, range 28-76) along with SLNs is shown in Table II. For each, histopathological staging (pT, pN) and tumour grading (G) are indicated. Lymph node levels with nodes enhancing either with 99mTc or ICG were collected and the total number of lymph nodes identified was reported as ‘green and hot’ (ICG + Tc +), ‘green and cold’ (ICG + Tc -) and ‘white and hot’ (ICG - Tc +). Lastly, ‘white and cold’ (ICG - Tc -) refers to the nodes which were found in the residual levels dissected as traditional neck dissection requires (Tab. III).
Lastly for each patient, Table IV displays the following: the count of nodes removed after ICG-guided SND (i.e. the number of fluorescent SLNs), the total lymph node count after bilateral modified radical ND (I-V levels), and the count and level of any metastatic lymph nodes.
An average of 6.9 (range 2-16) ‘green and hot’ lymph nodes (n = 62, ICG + Tc +) and of 7.8 (range 2-18) ‘green and cold’ lymph nodes (n = 70, ICG + Tc -) were identified. Almost all radioactive lymph nodes seen on lymphoscintigraphy were also visible with ICG fluorescence, as in the whole study only a single “white and hot” node was not detected with ICG (n = 1, ICG - Tc +). The number of green lymph nodes (ICG +) identified was higher than the hot ones (Tc +) but all were in the same lymph node level.
We calculated the sensitivity of the ICG method equal to 100%. A PPV of 47% compared to lymphoscintigraphy with 99mTc and 6.3% compared to lymph nodes with metastatic cells alone and an NPV of 100% in both cases, as ICG identified all hot and metastatic lymph nodes, were found.
The Robbin’s levels most frequently involved by enhancing SLNs were IB, IIA and III (83%), followed by level IV (66%). Moreover, enhancing nodes were detected in 50% of cases in level IIB and in 33% in level IA. No SLNs were located at level V. In 3 patients, a contralateral tumour lymphatic drainage was discovered. More in details, in patient #1 a contralateral diffusion in levels IIA and III was encountered, while in patient #4 the same result appeared in levels III-IV and in patient #5 in levels IA-IB-IIA.
We did not observe any enhancing lymph node (fluorescent and/or radioactive) in the T-N tract, the region connecting the primary tumour to the neck lymph nodes. After super-selective ICG guided ND of the enhancing SLNs, in all patients neck dissection was then completed with bilateral modified radical ND (levels I-V), removing an average total number of 56.3 lymph nodes (range 22-104).
The histopathological examination confirmed the clinical stage (cT4-pT4, according to the 8th edition of the TNM classification) in all cases. In 3 patients, lymph node metastases were detected despite negative findings within the preoperative clinical-radiological evaluation (cN0/pN+). All metastatic nodes were identified by both methods (green, ICG + and hot, Tc +); however, during neck dissection, precise localization was achievable solely through visual reference to the fluorescence of ICG.
All patients received postoperative adjuvant treatment with radiotherapy, potentially combined with chemotherapy based on pathological staging, in accordance with international guidelines.
Throughout the 43-month follow-up period (range 24-85 months), one patient developed lung and mediastinal lymph node metastases, and another underwent right upper lobectomy for lung adenocarcinoma. No patient experienced loco-regional relapse.
Discussion
Cervical metastasis in oral squamous cell carcinoma serves as a significant prognostic indicator. Therefore, precise diagnosis and treatment of potential occult neck disease are pivotal for optimising survival outcomes 1.
Currently, amongst numerous debates and in alignment with the 2018 AJCC guidelines, END is considered a viable option for early-stage tumours, while it is deemed mandatory for advanced T categories. The determination of the extent of neck dissection is guided by statistical analyses focusing on nodal levels presenting a high risk for tumour metastases 4,6.
The controversies surrounding this topic, along with cases of skip metastases observed in clinical practice and reported in the literature (whether contralateral or in level IV), highlight the complex and variable lymphatic drainage patterns in the oral cavity.
In 2006, De Cicco and colleagues published a paper regarding the lymphatic mapping of the neck by preoperative lymphoscintigraphy in 14 patients affected by locally advanced cN0 oral tongue cancer 14. Their results demonstrated that lymphoscintigraphy was always successful in detecting the lymphatic drainage. They also highlighted an otherwise unexpected bilateral drainage in 11 of 14 cases, contradicting the traditional concept of metastatic progression from level I to subsequent levels. The authors concluded that scintigraphy can reveal the functional anatomy of lymphatic drainage on a case-by-case basis, enabling the surgeon to plan ND based on such preoperative lymphatic mapping. However, we argue that a major limitation of relying solely on preoperative lymphoscintigraphy is the lack of real-time intraoperative visual feedback.
Recently, a new technique based on the observation of ICG NIR fluorescence for intraoperative lymphatic mapping showed good prospectives 13.
This method has the advantage of allowing real-time visual recognition, without altering the operative field and without exposing individuals to any radiation. ICG is a low molecular-weighted, non-toxic dye cleared quickly via a hepatic route. The initial validation of ICG feasibility in head and neck cancer was accomplished by Bredell et al. over a decade ago, despite the utilisation of different techniques and the examination of tumours with diverse origin (oropharyngeal SCC) 15.
Ishiguro et al. combined preoperative CT lymphography with intraoperative ICG injection for SLN detection in early-stage cN0 oral tongue cancers 16. The combined technique allowed the authors to detect SLN in 96.3% of cases in a series of 27 patients, most of which were located at ipsilateral level IB (68.3%) 16.
In the present pilot study, all the lymph nodes identified by lymphoscintigraphy were also successfully visualised by observation of ICG NIR fluorescence, resulting in a 100% identification rate.
The fluorescence lymphography with ICG exhibited a sensitivity and an NPV of 100% in comparison to the known values of the current reference method and the histopathological outcome. Nevertheless, despite the fact of being identified in the same lymph node levels, the number of green SLNs (ICG +) identified was found to be higher than that of the hot ones (99mTc +). Similar results were found in a clinical trial comparing the use of ICG and lymphoscintigraphy using 99mTc in breast cancer, where ICG was identified in 215 of 220 dissected nodes and radiotracer was identified in 172 of the nodes 17. This observation underscores a key difference in the spatial resolution of the 2 techniques. As reported by the European Association of Nuclear Medicine Oncology Committee and European Sentinel Node Biopsy Trial Committee in 2009, at a typical depth of 30 mm, a point-like lymph node is visualised as having an apparent diameter of approximately 25 mm, reflecting the intrinsic spatial resolution limits of the probe 18. Moreover, Britten in 1998 estimated a probe depth error of ± 0.5 mm and found that even the best performing probe required at least 51 mm separation to allow detection from the injection site 19. Hence, lymphoscintigraphy might not distinguish lymph nodes that are in close proximity (e.g., within a nodal cluster), whereas ICG fluorescence, providing real-time mapping in the open surgical field, allows a more precise identification of each individual lymph node. Nevertheless, through the use of this new personalised ND, we obtained a higher diagnostic accuracy of 4.8 percentage points, albeit in a minimal sample.
In the management of cN0 tongue cancers, the SLB technique has emerged as an alternative to END, gaining increasing popularity over the past decade. The concept of selective node biopsy in tongue cancer involves identification, removal, and analysis of the primary echelon lymph node in the tumor’s drainage network to detect micrometastases. Consequently, only the positive cases require further surgical intervention, namely END 21.
The use of SLB within the management of oral cancer is quite limited and still under debate. However, in terms of patient’s quality of life and cost-effectiveness its oncological role can be reinforced by numerous studies 23-25. In a meta-analysis of 3566 sentinel node biopsies, Liu et al reported 87% sensitivity and 94% NPV 24. In the French Senti-MERORL trial 307 cT1-2N0 were randomised between END and SLB. After a mean follow-up of almost 5 years, the 2- and 5-year nodal recurrence-free survival for END and SLB were comparable: 89.6% vs 90.7% and 89.6% vs 89.4%, respectively 25. Furthermore, locoregional recurrence free, disease-specific and overall survival did not differ between the 2 groups. Consequently, a significant number of studies comparing END to SNB have not included further comparisons involving patients who required a second surgery due to a positive sentinel node biopsy. This represents the main disadvantage of the sentinel biopsy strategy. Indeed, it is of relevance to view sentinel biopsy as a diagnostic surgical procedure, with the understanding that a significant number of patients may require a second neck surgery. This carries a higher risk of complications compared to upfront END in a surgical field that has not been previously operated on. In contrast to the standard sentinel node biopsy, our study departs from the conventional approach and introduces a completely alternative strategy for managing the cN0 neck in a specific oral cancer category. The present approach constitutes a single diagnostic and therapeutic procedure, thereby obviating the necessity for a traditional END as well an eventual additional surgery. The main advantage is to reduce postoperative morbidity and costs while maintaining comparable oncological outcomes for the patient. This can be achieved by removing not only the sentinel node, but also the entire enhancing lymphatic network responsible for draining the mucosal area of the primary tumour (i.e. tongue cancer in the present series).
Our results demonstrate that lymphoscintigraphy and ICG were always successful in detecting lymphatic drainage, even in bilateral cases. ICG supplied intraoperative information, thus completing scintigraphic data. The ICG method demonstrated high sensitivity (100%), indicating its ability to correctly identify the lymphatic drainage, as all metastatic nodes have been diagnosed exclusively in radiocolloid and ICG positive levels, while the non-tracer positive stations were free from disease. However, the PPV concerning metastatic lymph nodes was relatively low, at 6.3%. This suggests that, while the ICG test may be useful for detecting all lymph nodes potentially involved by metastasis, its ability to confirm the actual presence of metastasis is limited.
The aim of our pilot study was to establish tumour-related neck lymphatic mapping by evaluating the feasibility and reliability of the intraoperative fluorescence method in a cohort of patients with advanced stage cN0 oral tongue squamous cell carcinoma. The technique presented seeks to overcome the limitations of lymphoscintigraphic guided sentinel node biopsy by integrating it with ICG guidance. The intraoperative use of ICG enables real-time visualisation of the lymphatic stream, facilitating a personalised ICG-guided neck dissection targeting both the sentinel node and the connecting lymphatic network. We hypothesise that a case-specific lymphatic mapping approach could offer a more comprehensive strategy for managing the neck in patients with locally advanced cN0 oral disease. This approach would focus on limiting the extent of neck dissection to the levels directly affected by the disease while also addressing clinically unexpected skipped or contralateral drainage patterns.
Our results demonstrate the efficacy of using ICG in detecting patient-specific lymphatic streams and occult metastases. Nevertheless, the present study has limitations. Firstly, the sample size was insufficient for a comprehensive statistical evaluation to determine the most affected levels in oral tongue cancer, partly due to challenges in identifying eligible patients for enrollment. Additionally, the financial implications of procedural costs, including the additional time required for surgical procedure and the necessary equipment which might not be available in all clinics, must be taken into account. Based on our clinical experience, the use of ICG fluorescence visualisation through endoscopic instruments significantly enhances surgical dissection during sentinel lymph node biopsy procedures. Such improved manoeuvrability guarantees more precise localisation and dissection of lymphatic structures, ultimately contributing to reduced operative time and improved workflow efficiency.
Further studies are required to confirm the diagnostic accuracy of lymphatic mapping using ICG fluorescence and validate the innovative method proposed herein, with a larger sample size.
Conclusions
This study introduces a novel technique that has the potential to significantly impact the management of cN0 oral squamous cell carcinoma. The combination of preoperative lymphoscintigraphy with 99mTc-labeled colloids and intraoperative NIR fluorescence imaging using ICG facilitate the identification of cervical nodal stations and the lymphatic network at higher risk of metastases in cN0 tongue cancer. Furthermore, they are able to detect any individual variations in lymph node distribution for the same primary tumour site. The detection of occult metastases confined to the identified lymph node stations suggests that selective, tailored, and lymphography-guided neck dissection could overcome the limitations of sentinel biopsy. This approach may also reduce surgical complications and avoid the pitfalls associated with traditional elective neck treatment in oral cavity cancers. Further prospective studies on larger patient cohorts are required to validate these results.
Conflict of interest statement
The authors declare no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author contributions
All authors should have made substantial contributions to all of the followings: LC, MT, RB, MA: conceptualization; LC, MT, RB, VN, VC, Mab: methodology; LC, MT, MA: validation; LC, MT, RB, VN, VC: investigation; LG, EF, GR, Mab: data curation; LG, EF, GR: writing - original draft; GR, Mab: writing - review&editing; LC, MA, Mab: supervision; VN, MA: project administration. All of the authors have read and approved the manuscript.
Ethical consideration
This study was approved by the Institutional Ethics Committee (IEO Ethics Committee) (protocol number cod. IEO S629/411).
The research was conducted ethically, with all study procedures being performed in accordance with the requirements of the World Medical Association’s Declaration of Helsinki.
Written informed consent was obtained from each participant/patient for study participation and data publication.
History
Received: December 6, 2024
Accepted: September 2, 2025
Figures and tables
Figure 1. Preoperative detection of SPECT/CT lymphoscintigraphic enhancing lymph node.
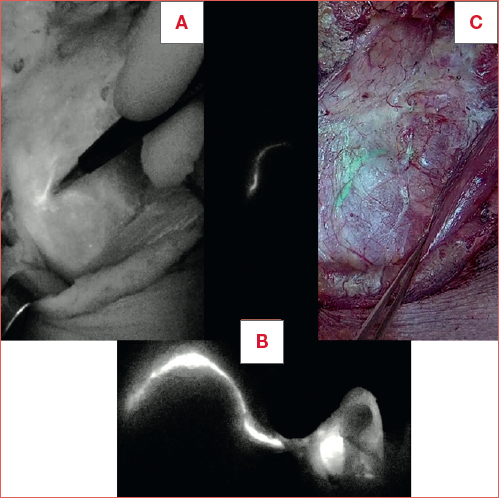
Figure 2. Intraoperative ICG procedure. A) Incision and preparation of subplatysmal flap; B) submucosal injection of ICG in 4 cardinal peritumoral points; C) visualization of the lymphatic network; D) removal of the ICG drainage tract and lymph node.
| Gold standard + | Gold standard - | |
|---|---|---|
| ICG + | A | B |
| ICG - | C | D |
Table II. Staging, visual characteristics of neck dissections and number of bi-modality enhancing lymph nodes of the cohort. ICG: indocyanine green; Tc: radiocolloid 99mTc.
Table III. Number of lymph nodes removed per level according to the Robbins classification. ICG: indocyanine green; Tc: radiocolloid 99mTc
| ICG neck dissection | Selective neck dissection | Metastatic lymph nodes | |
|---|---|---|---|
| Case 1 | 34 | 53 | 4 |
| Case 2 | 20 | 104 | 0 |
| Case 3 | 16 | 46 | 1 |
| Case 4 | 8 | 88 | 0 |
| Case 5 | 12 | 44 | 0 |
| Case 6 | 14 | 71 | 1 |
| Case 7 | 6 | 22 | 2 |
| Case 8 | 8 | 38 | 0 |
| Case 9 | 16 | 41 | 0 |
References
- Cerezo L, Millán I, Torre A, et al. Prognostic factors for survival and tumor control in cervical lymph node metastases from head and neck cancer. A multivariate study of 492 cases. Cancer 69:1224-1234, 1992. https://doi.org/10.1002/cncr.2820690526
- Hutchison IL, Ridout F, Cheung SMY, et al. Nationwide randomised trial evaluating elective neck dissection for early stage oral cancer (SEND study) with meta-analysis and concurrent real-world cohort. Br J Cancer 2019;121:827-836. https://doi.org/10.1038/s41416-019-0587-2
- Greenberg JS, El Naggar AK, Mo V, et al. Disparity in pathologic and clinical lymph node staging in oral tongue carcinoma. Implication for therapeutic decision making. Cancer 2003;98:508-515. https://doi.org/10.1002/cncr.11526
- Abu-Ghanem S, Yehuda M, Carmel N-N, et al. Elective neck dissection vs observation in early-stage squamous cell carcinoma of the oral tongue with no clinically apparent lymph node metastasis in the neck: a systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg 2016;142:857-865. https://doi:10.1001/jamaoto.2016.1281
- Samant S, Robbins KT. Evolution of neck dissection for improved functional outcome. World J Surg 2003;27:805-810. https://doi.org/10.1007/s00268-003-7113-6
- Shah JP, Andersen PE. The impact of patterns of nodal metastasis on modifications of neck dissection. Ann Surg Oncol 1994;1:521-532. https://doi.org/10.1007/BF02303619
- Shah JP, Candela FC, Poddar AK. The patterns of cervical lymph node metastases from squamous carcinoma of the oral cavity. Cancer 1990;66:109-113. https://doi.org/10.1002/1097-0142(19900701)66:1<109::aid-cncr2820660120>3.0.co;2-a.
- Ferreli F, Festa BM, Costantino A, et al. Prevalence of occult level 2b nodal metastases in cN0 squamous cell carcinoma of the oral cavity: a systematic review and meta-analysis. Oral Oncol 2021;122:105540. https://doi.org/10.1016/j.oraloncology.2021.105540
- Feng Z, Li JN, Niu LX, et al. Supraomohyoid neck dissection in the management of oral squamous cell carcinoma: special consideration for skip metastases at level IV or V. J Oral Maxillofac Surg 2014;72:1203-1211. https://doi.org/10.1016/j.joms.2013.12.008
- Holden AM, Sharma D, Schilling C, et al. Biopsy of the sentinel lymph node in oral squamous cell carcinoma: analysis of error in 100 consecutive cases. Br J Oral Maxillofac Surg 2018;56:615-620. https://doi.org/10.1016/j.bjoms.2018.06.019
- Galimberti V, Cole BF, Viale G, et al. Axillary dissection versus no axillary dissection in patients with breast cancer and sentinel-node micrometastases (IBCSG 23-01): 10-year follow-up of a randomized, controlled phase 3 trial. Lancet Oncol 2018;19:1385-1393. https://doi.org/10.1016/S1470-2045(18)30380-2
- Khafif A, Schneebaum S, Fliss DM, et al. Lymphoscintigraphy for sentinel node mapping using a hybrid single photon emission CT (SPECT)/CT system in oral cavity squamous cell carcinoma. Head Neck 2006;28:874-879. https://doi.org/10.1002/hed.20434
- Honda K, Ishiyama K, Suzuki S, et al. Sentinel lymph node biopsy using preoperative computed tomographic lymphography and intraoperative indocyanine green fluorescence imaging in patients with localized tongue cancer. JAMA Otolaryngol Head Neck Surg 2019;145:735-740. https://doi.org/10.1001/jamaoto.2019.1243
- De Cicco C, Trifirò G, Calabrese L, et al. Lymphatic mapping to tailor selective lymphadenectomy in cN0 tongue carcinoma: beyond the sentinel node concept. Eur J Nucl Med Mol Imaging 2006;33:900-905. https://doi.org/10.1007/s00259-006-0088-4
- Bredell MG. Sentinel lymph node mapping by indocyanin green fluorescence imaging in oropharyngeal cancer – Preliminary experience. Head Neck Oncol 2010;2:31. https://doi.org/10.1186/1758-3284-2-31
- Ishiguro K, Iwai T, Izumi T, et al. Sentinel lymph node biopsy with preoperative CT lymphography and intraoperative indocyanine green fluorescence imaging for N0 early tongue cancer: a long-term follow-up study. J Craniomaxillofac Surg 2020;48:217-222. https://doi.org/10.1016/j.jcms.2020.01.007
- Papathemelis T, Jablonski E, Scharl A, et al. Sentinel lymph node biopsy in breast cancer patients by means of indocyanine green using the Karl Storz VITOM® Fluorescence Camera. Biomed Res Int 2018:6251468.
- Alkureishi LW, Burak Z, Alvarez JA, et al. European Association of Nuclear Medicine Oncology Committee; European Sentinel Node Biopsy Trial Committee. Joint practice guidelines for radionuclide lymphoscintigraphy for sentinel node localization in oral/oropharyngeal squamous cell carcinoma. Eur J Nucl Med Mol Imaging 2009;36:1915-1936. https://doi.org/10.1007/s00259-009-1248-0
- Britten AJ. A method to evaluate intra-operative gamma probes for sentinel lymph node localisation. Eur J Nucl Med 2019;26:76-83. https://doi.org/10.1007/s002590050362
- De Bree R, de Keizer B, Civantos FJ, et al. What is the role of sentinel lymph node biopsy in the management of oral cancer in 2020? Eur Arch Otorhinolaryngol 2021;278:3181-3191. https://doi.org/10.1007/s00405-020-06538-y
- Gurney BA, Schilling C, Putcha V, et al. Implications of a positive sentinel node in oral squamous cell carcinoma. Head Neck 2012;34:1580-1585. https://doi.org/10.1002/hed.21973
- Mahieu R, den Toom IJ, Boeve K, et al. Contralateral regional recurrence in lateralized or paramedian early-stage oral cancer undergoing sentinel lymph node biopsy – Comparison to a historic elective neck dissection cohort. Front Oncol 2021;11:644306. https://doi.org/10.3389/fonc.2021.644306
- Civantos FJ, Zitsch RP, Schuller DE, et al. Sentinel lymph node biopsy accurately stages the regional lymph nodes for T1-T2 oral squamous cell carcinomas: results of a prospective multiinstitutional trial. J Clin Oncol 2010;28:1395-1400. https://doi.org/10.1200/JCO.2008.20.8777
- Liu M, Wang SJ, Yang X, et al. Diagnostic efficacy of sentinel lymph node biopsy in early oral squamous cell carcinoma: a meta-analysis of 66 studies. PLoS One 2017;12:E0170322. https://doi.org/10.1371/journal.pone.0170322
- Garrel R, Poissonnet G, Moyà Plana A. Equivalence randomized trial to compare treatment on the basis of sentinel node biopsy versus neck node dissection in operable T1-T2N0 oral and oropharyngeal cancer. J Clin Oncol 2020;38:34,4010-4018. https://doi.org/10.1200/JCO.20.01661
Downloads
License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright
Copyright (c) 2026 Società Italiana di Otorinolaringoiatria e chirurgia cervico facciale
How to Cite
- Abstract viewed - 576 times
- PDF downloaded - 166 times



