Rhinology
Vol. 46: Issue 2 - April 2026
Endoscopic transnasal craniectomy: impact on staging and prognosis of sinonasal adenocarcinoma
Summary
Objectives. Postoperative prognosis of sinonasal adenocarcinoma (SNAC) is closely related to local recurrence. Our objective was to identify subclinical anterior skull base (ASB) invasion, and to evaluate the impact of endoscopic transnasal craniectomy on SNAC staging and prognosis.
Methods. Retrospective study conducted from 2010 to 2023, including 33 patients, comparing 2 groups: conservative endoscopic sinus surgery (ESS) and complementary endoscopic resection with transnasal anterior skull base craniectomy (ERTC). Histopathology, overall survival (OS), recurrence-free survival (RFS), complication rates, surgery time and hospital stay were analysed.
Results. Histopathology revealed malignant invasion in 36.8% of ASB surgical specimens, despite the absence of pre- or intraoperative evidence of skull base invasion. All were low-grade tumours, with 57.1% previously classified as cT2. ERTC was associated with an improvement in RFS, although not statistically significant, with a greater incidence of complications, but no impact on OS.
Conclusions. This study provides the first significant evidence of frequent subclinical invasion of the ASB in cT2 cases. These findings raise important questions regarding the management of cT2 lesions. Should systematic biopsies of the anterior skull base be taken? Should a skull base resection be discussed in selected patients? Further studies with larger samples are necessary to better define the risk-benefit ratio of ERTC in low-stage SNAC prognosis.
Introduction
Adenocarcinoma represents about 5 to 30% of all sinonasal tumours 1. Its incidence rate and histological distribution varies widely between countries 2. Most sinonasal adenocarcinomas (SNAC) are related to the occupational exposition to a volatile carcinogenic compound, wood dust being the most common 3–5. Mean age at diagnosis is 65 years and there are more cases in males, which is related to the occupational risk factors 6,7. The tumour originates in the olfactory cleft 8.
During last decades, treatment options have evolved towards less invasive procedures. The therapeutic gold standard is endoscopic surgical resection of the tumour, with adjuvant radiotherapy for tumour stages up to pT2 and/or high grade and/or incomplete resection 9-11. Prognosis is mainly based on initial tumour, nodes, and metastases (TNM) staging and local recurrence; lymph node involvement and distant metastases are very uncommon 1,12. Indeed, 88% of recurrences are local 13, frequently located in the anterior skull base (ASB) area and in the posterior ethmoid, even in low-stage tumours 14. Some authors suggested that total exenteration of the olfactory cleft may help grading the invasion and predict the risk of local recurrence 15.
In case of tumour invasion of the ASB, a radical resection is recommended, when considered reasonable and surgically feasible 16. However, there is no consensus regarding the resection of the ASB when preoperative imaging shows no skull base invasion. Some authors have suggested that a more radical attitude regarding the ASB could improve the prognosis 17,18.
The aim of our study was then to assess the prevalence of microscopic malignant invasion of the ASB in SNAC depending on the tumour characteristics, in patients without radiologic evidence of ASB invasion. The primary outcome was the definitive histopathology on ASB specimens. Secondary outcomes were surgical complication rate and malignant recurrence location.
Methods
Study design and data collection
Pseudonymised data of screened patients related to their characteristics, tumour, treatment, perioperative events, and follow-up were collected retrospectively from 2011 to 2023. Patients screened were adults (> 18 years-old) treated by endoscopic resection for histologically proven SNAC in Besançon University Hospital. Non-inclusion criteria were: invasion of the ASB on preoperative imaging; incomplete preoperative imaging protocol (absence of cervicothoracic computed tomography [CT] scan and/or magnetic resonance imaging [MRI] with gadolinium of sinonasal cavities); non-carcinologic resection; transcranial resection of the skull base; absence or refusal of radiotherapy when indicated (i.e., stage > T2 tumours, high grade, and/or incomplete resection); adjuvant or neoadjuvant chemotherapy; and lack of consent. The indications for adjuvant radiotherapy were based on the recommendations of the Réseau d’expertise français sur les cancers ORL rares (REFCOR). Barnes’ histopathologic classification was used for tumour grading. All cases were re-assessed for preoperative TNM staging using Union for International Cancer Control (UICC) 2017 classification. All CT and MRI images were re-interpreted with contribution from a head and neck specialist radiologist. All eligible patients who underwent endoscopic resection with transnasal craniectomy (ERTC) were included. A control group of patients who underwent endoscopic sinus surgery without resection of the skull base (ESS) was also included to compare the characteristics of patients, tumours, and recurrences.
Surgical technique and follow-up
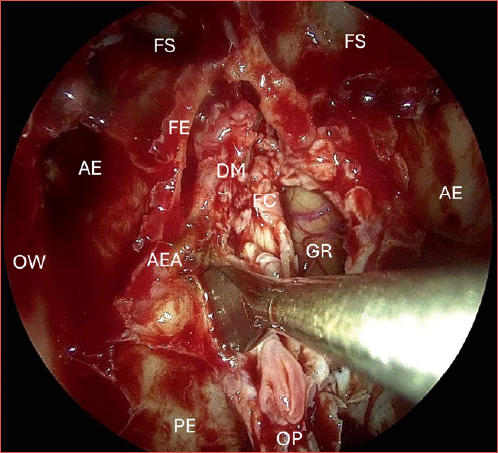
All patients underwent a fully endoscopic bilateral ethmoidectomy with sphenoidostomy and septectomy, resection of the middle turbinate, conchal lamina and complete exenteration of the olfactory cleft, with or without transnasal anterior skull base resection. When performed, endoscopic transnasal anterior craniectomy consisted in the resection of the entire lamina cribrosa, lateral lamella, crista galli, and overlying dura mater, as shown in the Cover figure. Abdominal fat graft with or without rectus abdominis fascia graft was used for the anterior skull base reconstruction. In cases with involvement of the medial part of the maxillary sinus, a medial maxillectomy was added to the procedure. The decision to resect the ASB was made prior to surgery according to the surgeon’s standard practice and the recommendation of the regional multidisciplinary oncology board. However, in some cases, intraoperative findings changed the approach. Evidence of tumour lysis or positive frozen sections on the skull base led to the resection of the ASB, when not previously anticipated. The retrospective determination of whether frozen sections were performed as a systematic margin assessment or in response to suspicion of tumour invasion was not possible. Therefore, these cases were excluded from the analyses. Thus, the study only included cases that were considered radiologically, clinically and surgically free of tumour involvement of the skull base.
The follow-up protocol was based on national guidelines established by the REFCOR and consisted of clinical examinations with nasal fibroscopy at 3-month intervals during the first 2 years, 4-month intervals during the third year, 6-month intervals during the fourth and fifth years, and annual examinations thereafter. An MRI of the face was conducted every 6 months during the initial 5-year period, after which it was performed annually.
Statistical analysis
Overall survival (OS) and recurrence-free survival (RFS) were evaluated with Kaplan-Meier method and compared using log-rank test. Characteristics of the 2 groups and secondary outcomes were compared using Fisher’s test for qualitative variables and 2-tailed T-test for quantitative variables. Significance level was set at p < 0.05. All statistical analysis were performed with R (4.1.2 version, R Core Team, 2017, R Foundation for Statistical Computing, Vienna, Austria).
Ethics and regulatory aspects
Our work was in accordance with national regulatory instances regarding the use of personal and medical data. As required by ethics committees, all eligible patients received an information and consent note with a refusal form.
Results
Population
In our database, 81 patients were treated for SNAC from 2011 to 2021. A total of 47 patients who underwent full endoscopic resection was evaluated. Thirty-five met the inclusion criteria. Two patients who underwent only partial resection of the skull base were excluded from the study. Ultimately, 33 patients were considered for statistical analyses. Among these, 14 (42.4%) patients had a conservative carcinologic ESS, while 19 (57.6%) had an ERTC. None had lymph node involvement or distant metastases at the time of diagnosis. The 2 groups were compared for all population characteristics, showing no significant differences (Tab. I). Twelve patients (36.4%) presented a locally advanced tumour (pT3-T4). One well-differentiated, low-grade non-intestinal SNAC (n-ITAC) was included. Thirty patients (90.9%) had adjuvant radiotherapy, the 3 others presented a low-grade pT1 or pT2 tumor without indication of adjuvant radiotherapy.
Primary outcome
Definitive histopathologic analysis revealed that 7 (36.8%) craniectomy specimens exhibited microscopic malignant invasion of the ASB in the ERTC group. All were histologically low-grade tumours. Clinical tumour staging of these cases was: 4 T2 (57.1%), 2 T3 (28.6%) and one T4a (14.3%). All these patients had adjuvant radiotherapy and none presented tumour recurrence.
Secondary outcomes
Ten (30.3%) patients presented a recurrence, 6 (42.9%) of the ESS group and 4 (21.1%) of the ERTC group as shown in Table II. All were local recurrences, associated with cervical and mediastinal lymph node metastases in one case. One recurrence in the frontal lobe was found in the ERTC group (5.3%), whereas 4 patients (28.6%) from the ESS group presented a recurrence involving the ASB with or without intracranial extension. Among these 5 patients, 2 necessitated surgical revision by craniofacial resection, the other 3 presented a locally advanced lesion not eligible for surgery and were treated by chemotherapy or palliative measures.
Eight patients (24.2%) presented a total of 11 postoperative complications (Tab. III). None led to death. The number of complications was higher in the ERTC group (p = 0.037). Specifically, the ERTC group exhibited a greater incidence of mild complications than the ESS group (p = 0.42), while the difference of severe complications was not statistically significant (p = 0.162). The most common complications were cerebrospinal fluid leak in 4 cases (12.1%) and meningitis in 2 cases (6.1%). Median time of surgery was 5 hours and 10 minutes and was not statistically different between the 2 groups (p = 0.193). Median hospital stay length was 6 days for the entire sample and was longer in the ERTC group (7 days) than the ESS group (5 days) but not significantly (p = 0.055).
Discussion
Jankowski et al. demonstrated that the origin area of SNAC is the olfactory cleft 8. The prognosis of this cancer is closely related to local recurrence, notably in the anterior skull base 1,12. De Gabory et al. showed the serious consequences of a tumour invading the ASB, particularly in the case of a meningeal invasion 19. In this case, surgical resection of the ASB is necessary 16. Some authors have proposed a resection of the ASB in case of a tumour located on or close to the lamina cribrosa 10,17. Considering this, we hypothesised that adopting a more radical approach to the anterior skull base might prove beneficial in terms of tumour control. In previous literature, it was not possible to investigate the impact of a transnasal craniectomy on adenocarcinomas limited to the sinonasal cavities, as this surgical procedure was typically performed for patients with skull base invasion.
Our sample was representative of the population presenting a SNAC described in the literature 9. The proportion of T3-T4 cases was lower, knowing that we excluded locally advanced tumours invading the ASB (corresponding to T3, T4a or T4b stages).
We have demonstrated that 36.8% of ASB surgical specimens in the ERTC group exhibited malignant invasion, despite the absence of radiological or intraoperative evidence of tumour invasion of the skull base. Furthermore, these subclinical invasions were not correlated with high cT stages, with 57.1% of cases previously classified as cT2. To the best of our knowledge, the present study is the first to prove the frequent malignant invasion of the ASB in patients who have not demonstrated any radiological or clinical evidence of such invasion. Our study supports that preoperative imaging has a poor concordance with histopathology, resulting in an underestimation of the cT staging. These findings led to a frequent upgrading of the pT status and consequently influenced the decision regarding adjuvant therapy. Indeed, current REFCOR guidelines recommend an adjuvant radiotherapy for tumour stages up to T2 and/or high grade and/or incomplete resection. This seems consistent with the results of Camp et al. 14 who observed 24% positive margins on olfactory phyla in definitive histopathologic analysis. In contrast, Fierens et al. 20 pointed out that preoperative imaging frequently led to an overestimation of the cT staging. The authors report that 69% of the pT1-pT2 cases underwent resection of the skull base, including situations where it could have been avoided. It still can be hypothesised that in certain cases with surgical preservation of the ASB, a microscopic invasion of the skull base might be ignored.
This finding raises the question of adjuvant radiotherapy for patients with pT2 tumours who have undergone surgical preservation of the anterior skull base. Interestingly, Turri-Zanoni et al. 11 found that adjuvant radiotherapy did not significantly improve the prognosis of pT2 cases. However, they observed better survival trends in the radiotherapy group. The authors concluded that adjuvant radiotherapy could still be suggested for this population, although there is no strong evidence of its beneficial effects. Further studies are required to investigate the impact of radiotherapy in this specific population.
In the study by Camp et al. 14, similar locations of recurrence than ours were found: 35% were located on the ASB and 45% in the posterior ethmoid. In this study, despite a “boost” of radiation therapy targeting the recurrence site, 38% presented a second recurrence, 54.5% of which being located on the ASB. Eventually, most necessitated a salvage therapy by craniofacial resection. The author explained the high number of local recurrences by a possible insufficient resection of the ASB, a hypothesis that was also supported by Nicolai et al. 17.
Complication rate was consistent with the rates in other studies (Tab. IV) and was significantly higher in the ERTC group, with none being lethal. In the literature, it appears that this rate seems to be correlated with the proportion of transnasal craniectomies. Indeed, the highest number of complications is found in the study by Oker et al. who performed ERTC on 85% of the population 21. Nicolai et al. found a statistically significant difference, but the analysis included a transcranial resection group 17. In the latter study, hospital stay was longer in the case of craniectomy (ESS, 7.4 days and ERTC, 14.1 days). In our work, there was also a difference of hospital stay length but not in significant proportions. On the contrary, Vergez et al. did not find any significant difference in complications or hospital stay length depending on the resection of the ASB 11. These data suggest that patients should be carefully selected for transnasal craniectomy depending on their comorbidities and functional status. As previously stated, adjuvant radiotherapy should be considered as an alternative treatment option, particularly in patients with significant comorbidities. Further studies would be beneficial in assessing the risk-benefit ratio of a systematic transnasal craniectomy versus systematic adjuvant radiotherapy in T2 tumours.
In our study, one well-differentiated, low-grade n-ITAC was included. Notwithstanding the significant histopathological discrepancy between intestinal SNAC (ITAC) and n-ITAC, the decision was taken to retain this case. This was done in accordance with the objectives of the study, which were to concentrate on microscopic invasion and prognosis in relation to the surgical technique. Both surgical techniques (ESS and ERTC) are suitable for n-ITACs, depending on their clinical presentation. Both tumour types have been observed to demonstrate a tendency to invade the anterior skull base 25. The existing body of literature provides equivocal information regarding the prognosis and the specific standard of care required for these tumours. The study by Chen et al. 26 reported no significant difference in survival when comparing 300 n-ITACs and 25 ITACs. Bhayani et al. 27 came to the same conclusion in their study involving 30 of 66 patients presenting n-ITAC. Conversely, Bignami et al. found better outcomes than those documented in the literature. They also highlighted that, as for ITACs, the ability to reach free-margins resection is the paramount prognostic factor. This conviction is also held by the present authors and formed the motivation for the present study.
This study had some limitations. The primary limitation of this study was the small sample size, which was a consequence of the low prevalence of this cancer and the monocentric nature of the study. Since there is no evidence that ERTC is superior to ESS in a given situation, the choice of surgical technique was left to the surgeon after a recommendation from a multidisciplinary oncology board. This can be viewed as a source of bias. However, different surgeons performed one technique or the other. This helped limit the risk of bias related to the surgeon’s experience. Finally, due to the rarity of SNAC and the necessity of a long follow-up period to detect long-term recurrences, a retrospective study design was chosen, despite the potential for bias. As a further development of this work, a multicentre study involving a much larger sample would be highly informative in evaluating the generalisability of our results and assess the prognostic impact of a systematic ERTC. In addition, a comprehensive evaluation of the overall survival and the recurrence-free survival, in conjunction with an assessment of the quality of life associated with the surgical technique, would facilitate a more comprehensive understanding of the tangible impact of the surgery and its complications.
Conclusions
This study provides the first significant evidence of a frequent subclinical tumour invasion of the anterior skull base in small stage cT2 SNAC cases, thus confirming the poor radiologic-pathologic correlation. These findings enabled a reassessment of the TNM classification, which impacted the indications of adjuvant radiotherapy. Our results indicate that systematic biopsies of the anterior skull base, perhaps involving a more radical resection, might help to prevent misdiagnosis and to treat subclinical invasion of the skull base. Adjuvant radiotherapy should be considered to achieve local control of the primary tumour site in T2 lesions, particularly when a transnasal craniectomy is not deemed a reasonable option. Further studies with larger sample sizes are necessary to more fully define the risk-benefit ratio of systematic transnasal craniectomy on the prognosis of SNAC.
Acknowledgements
We thank Dr. Zakia Boulahdour for her valuable contribution to the review of patients’ imaging.
Conflict of interest statement
The authors declare no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author contributions
JM, OM: investigation, drafting of the manuscript; All authors: conceptualisation, methodology, review and editing, validation; JM: formal analysis.
Ethical consideration
This retrospective study did not involve human subjects and therefore did not fall within the scope of the Jardé law, in compliance with the law on research involving the human person (RIPH) n° 2012-300 of March 5, 2012, implementing decree n° 2016-1537 of November 16, 2016, order n° 2016-800 of June 16, 2016 amending chapter I of title II of book 1 of the public health code relating to RIPH, decree n° 2017-884 of May 9, 2017 amending certain provisions relating to RIPH and the orders in force. Consequently, all eligible patients received an information note with a refusal form.
This work was in accordance with the MR-004 reference methodology established by the French National Data Protection Authority (CNIL) regarding the use of personal and medical data.
History
Received: December 19, 2024
Accepted: December 15, 2025
Figures and tables
| Characteristics | All, N = 33 | ESS, N = 14 | ERTC, N = 19 | p value |
|---|---|---|---|---|
| Gender | .496 | |||
| Female | 2 (6.1) | 0 | 2(11) | |
| Male | 31(94) | 14(100) | 17(89) | |
| Age at diagnosis | 70 (62, 79) | 70 (64, 74) | 68 (62, 80) | .662 |
| Laterality | .72 | |||
| Right | 13 (39.4) | 6 (42.9) | 7(37) | |
| Left | 19 (57.6) | 7(50) | 13(63) | |
| Bilateral | 1(3) | 1 (7.1) | 0 | |
| T stage (UICC 2017) | .16 | |||
| 1 | 3 (9.1) | 3(21) | 0 | |
| 2 | 18(55) | 6(43) | 12(63) | |
| 3 | 8(24) | 4(29) | 4(21) | |
| 4a | 4(12) | 1(7) | 3(16) | |
| Histology | .424 | |||
| Intestinal | 32(97) | 12(93) | 19(100) | |
| Non-intestinal | 1(3) | 1(7) | 0 | |
| Barnes’ classification | ||||
| Papillary | 13 (40.6) | 6(43) | 7(37) | .191 |
| Colonic | 7(22) | 4(29) | 3(16) | |
| Mucinous | 5 (15.6) | 1 (7.1) | 4 (21.1) | |
| Signet ring cells | 2 (6.3) | 1 (7.1) | 1 (5.3) | |
| Solid | 2 (6.3) | 2 (14.3) | 0 | |
| Mixed | 2 (6.3) | 0 | 2(11) | |
| Not specified | 3 (9.4) | 1 (7.1) | 3(16) | |
| Grade | ||||
| High grade | 7(21) | 3(21) | 4 (21.1) | .39 |
| Low grade | 23(70) | 11(79) | 12(63) | |
| Not specified | 3 (9.1) | 0 | 3(16) | |
| Adjuvant radiotherapy | 30 (90.9) | 13 (92.9) | 17 (89.5) | 1 |
| N (%); median (interquartile range). | ||||
| ESS: endoscopic sinus surgery; ERTC: endoscopic resection with transcranial craniectomy. | ||||
| Recurrences | All, N = 33 | ESS, N = 14 | ERTC, N = 19 |
|---|---|---|---|
| All, number of patients (%) | 10 (30.3) | 6 (42.9) | 4 (21.1) |
| Recurrence sites: | |||
| Anterior skull base and/or frontal lobe | 5 (15.2) | 4 (28.6) | 1 (5.3) |
| Posterior ethmoid and sphenoethmoidal recess | 4 (12.1) | 1 (7.1) | 3 (15.8) |
| Lamina papyracea | 1(3) | 1 (7.1) | 0 |
| N (%); ESS: endoscopic sinus surgery; ERTC: endoscopic resection with transcranial craniectomy. | |||
| Secondary outcomes | All, N = 33 | ESS, N = 14 | ERTC, N = 19 | p value |
|---|---|---|---|---|
| Patients presenting ≥ 1 complication | 8 (24.2) | 1 (7.1) | 7 (36.8) | |
| Complications | 11 | 1 | 10 | .037 |
| Mild complications: | 4 | 0 | 4 | .042 |
| CSF leak, medical treatment | 2 | 0 | 2 | |
| Meningismus | 1 | 0 | 1 | |
| Acute confusion | 1 | 0 | 1 | |
| Severe complications: | 7 | 1 | 6 | 0.162 |
| Meningitis | 2 | 1 | 1 | |
| Chronic subdural hematoma | 1 | 0 | 1 | |
| Chronic cerebral abscess | 1 | 0 | 1 | |
| Major pneumocephalus | 1 | 0 | 1 | |
| CSF leak, revision surgery | 2 | 0 | 2 | |
| Surgery time (hours: minutes) | 5:10 (3:56, 6:10) | 4:19 (3:31, 6:00) | 5:24 (4:06, 6:24) | .193 |
| Hospital stay duration (days) | 6 (5, 8) | 5 (4, 6) | 7 (6, 9) | .055 |
| N (%); median (interquartile range); bold values indicate significant results. | ||||
| CSF: cerebrospinal fluid; ESS: endoscopic sinus surgery; ERTC: endoscopic resection with transcranial craniectomy. | ||||
| Authors | Year | Country | No. of patients | Mean follow-up 1 | Recurrence rate (%) | Local rec. rate (%) | Median time to rec. 1 | Complication rate (%) |
|---|---|---|---|---|---|---|---|---|
| This study | 2023 | France | 33 | 75.7 | 30.3 | 30.3 | 34 | 24.2 |
| ESS | 14 | 42.9 | 42.9 | 34 | 7.1 | |||
| ERTC | 19 | 21.1 | 21.1 | 48 | 36.8 | |||
| De Gabory et al. a | 2022 | France | 257 | 52 | 24.6 | N/A | 36.8 | 8.4 |
| Russel et al. | 2018 | France | 67 | 61 | 23.9 | N/A | 41.5 | N/A |
| ESS | 56 | |||||||
| ERTCb | 11 | |||||||
| Schreiber et al. | 2018 | Italy | 27 | N/A | N/A | 7.4 | N/A | N/A |
| Oker et al. | 2017 | France | 60 | 34 | 21.6 | 13.3 | 10 | 33.3 |
| Camp et al. | 2016 | Belgium | 123 | N/A | N/A | 38 | 21 | N/A |
| Nicolai et al. a | 2015 | Italy | 169 | 42.8 | 21.3 | 16 | 13 | 17.1 |
| ESS | 38 | 7.9 | ||||||
| ERTCb | 103 | 11.7 | ||||||
| Vergez et al. | 2013 | France | 159 | 32.5 | 17.6 | N/A | 23 | 19 |
| ESS | 85 | 17.6 | 14.1c | |||||
| ERTC | 74 | 17.6 | 18.9c | |||||
| Van Gerven et al. | 2010 | Belgium | 44 | 61 | N/A | 36 | N/A | N/A |
| Bogaerts et al. | 2008 | Belgium | 44 | 36 | N/A | 27 | N/A | N/A |
| Lund et al. | 2007 | USA | 15 | N/A | N/A | N/A | N/A | N/A |
| Goffart et al. a | 2000 | Belgium | 78 | N/A | N/A | 30 | 12.8 | N/A |
| 1Time (months); aResults only for patients treated by full-endoscopic resection; b ERTC performed only for patients with tumoral invasion of the anterior skull base; cComplication rate calculated on 132 patients. | ||||||||
| ESS: endoscopic sinus surgery; ERTC: endoscopic resection with transcranial craniectomy; N/A: not available; OS: overall survival; rec.: recurrence, RFS: recurrence-free survival. | ||||||||
References
- Choussy O, Ferron C, Védrine P. Adenocarcinoma of ethmoid: a GETTEC retrospective multicenter study of 418 cases. Laryngoscope. 2008;118:437-443. doi:https://doi.org/10.1097/MLG.0b013e31815b48e3
- Rampinelli V, Ferrari M, Nicolai P. Intestinal-type adenocarcinoma of the sinonasal tract: an update. Curr Opin Otolaryngol Head Neck Surg. 2018;26:115-121. doi:https://doi.org/10.1097/MOO.0000000000000445
- Kleinsasser O, Schroeder H. Adenocarcinomas of the inner nose after exposure to wood dust. Arch Otorhinolaryngol. 1988;245:1-15. doi:https://doi.org/10.1007/BF00463541
- Bussi M, Gervasio C, Riontino E. Study of ethmoidal mucosa in a population at occupational high risk of sinonasal adenocarcinoma. Acta Otolaryngol. 2002;122:197-201. doi:https://doi.org/10.1080/00016480252814225
- Cantù G, Solero C, Mariani L. Intestinal type adenocarcinoma of the ethmoid sinus in wood and leather workers: a retrospective study of 153 cases. Head Neck. 2011;33:535-542. doi:https://doi.org/10.1002/HED.21485
- König M, Osnes T, Bratland Å. Treatment of sinonasal adenocarcinoma: a population-based prospective cohort study. J Neurol Surg B Skull Base. 2020;81:627-637. doi:https://doi.org/10.1055/s-0039-1694050
- De Gabory L, Conso F, Barry B. Carcinogenesis of the ethmoidal adenocarcinoma due to wood dust. Rev Laryngol Otol Rhinol (Bord). 2009;130:93-104.
- Jankowski R, Georgel T, Vignaud J. Endoscopic surgery reveals that woodworkers’ adenocarcinomas originate in the olfactory cleft. Rhinology. 2007;45:308-314.
- de Gabory L, Waubant A, Verillaud B. Multicenter study to assess surgical treatments of 452 sinonasal intestinal-type adenocarcinomas: a REFCOR study. Eur J Surg Oncol. 2023;49:39-46. doi:https://doi.org/10.1016/J.EJSO.2022.07.021
- Vergez S, du Mayne M, Coste A. Multicenter study to assess endoscopic resection of 159 sinonasal adenocarcinomas. Ann Surg Oncol. 2014;21:1384-1390. doi:https://doi.org/10.1245/s10434-013-3385-8
- Turri-Zanoni M, Battaglia P, Lambertoni A. Treatment strategies for primary early-stage sinonasal adenocarcinoma: a retrospective bi-institutional case-control study. J Surg Oncol. 2015;112:561-567. doi:https://doi.org/10.1002/JSO.24038
- Fiaux-Camous D, Chevret S, Oker N. Prognostic value of the seventh AJCC/UICC TNM classification of intestinal-type ethmoid adenocarcinoma: systematic review and risk prediction model. Head Neck. 2017;39:668-678. doi:https://doi.org/10.1002/hed.24663
- Cantù G, Bimbi G, Miceli R. Lymph node metastases in malignant tumors of the paranasal sinuses: prognostic value and treatment. Arch Otolaryngol Head Neck Surg. 2008;134:170-177. doi:https://doi.org/10.1001/ARCHOTO.2007.30
- Camp S, Van Gerven L, Vander Poorten V. Long-term follow-up of 123 patients with adenocarcinoma of the sinonasal tract treated with endoscopic resection and postoperative radiation therapy. Head Neck. 2016;38:294-300. doi:https://doi.org/10.1002/hed.23900
- Russel A, Nguyen D, Vigouroux C. Compartmentalized endoscopic resection of the olfactory cleft for nasal intestinal adenocarcinomas. Head Neck. 2018;40:2389-2398. doi:https://doi.org/10.1002/hed.25349
- Ferrari M, Bossi P, Mattavelli D. Management of sinonasal adenocarcinomas with anterior skull base extension. J Neurooncol. 2020;150:405-417. doi:https://doi.org/10.1007/s11060-019-03385-8
- Nicolai P, Schreiber A, Bolzoni Villaret A. Intestinal type adenocarcinoma of the ethmoid: outcomes of a treatment regimen based on endoscopic surgery with or without radiotherapy. Head Neck. 2016;38:E996-E1003. doi:https://doi.org/10.1002/HED.24144
- Vergez S, Martin-Dupont N, Lepage B. Endoscopic vs transfacial resection of sinonasal adenocarcinomas. Otolaryngol Head Neck Surg. 2012;146:848-853. doi:https://doi.org/10.1177/0194599811434903
- De Gabory L, Maunoury A, Maurice-Tison S. Long-term single-center results of management of ethmoid adenocarcinoma: 95 patients over 28 years. Ann Surg Oncol. 2010;17:1127-1134. doi:https://doi.org/10.1245/S10434-010-0933-3
- Fierens S, Moya-Plana A, Vergez S. Do practitioners assess sinonasal adenocarcinoma extension similarly? Interdisciplinary concordance in 21 cases. Clin Otolaryngol. 2021;46:665-669. doi:https://doi.org/10.1111/coa.13708
- Oker N, Verillaud B, Wassef M. Ethmoidal adenocarcinoma treated by exclusive endoscopic approach: focus on learning curve and modification of management. Head Neck. 2018;40:126-136. doi:https://doi.org/10.1002/hed.24990
- Huang E, Lu A, Tsai Y. Decreasing recurrence and increasing survival rates in patients of ethmoid or sphenoid intestinal-type adenocarcinomas: systematic review and meta-analysis with 1126 cases. Medicine. 2021;100. doi:https://doi.org/10.1097/MD.0000000000027341
- Bogaerts S, Vander Poorten V, Nuyts S. Results of endoscopic resection followed by radiotherapy for primarily diagnosed adenocarcinomas of the paranasal sinuses. Head Neck. 2008;30:728-736. doi:https://doi.org/10.1002/hed.20771
- Van Gerven L, Jorissen M, Nuyts S. Long-term follow-up of 44 patients with adenocarcinoma of the nasal cavity and sinuses primarily treated with endoscopic resection followed by radiotherapy. Head Neck. 2011;33:898-904. doi:https://doi.org/10.1002/hed.21556
- Bignami M, Lepera D, Volpi L. Sinonasal non–intestinal-type adenocarcinoma: a retrospective review of 22 patients. World Neurosurg. 2018;120:E962-E969. doi:https://doi.org/10.1016/j.wneu.2018.08.201
- Chen M, Roman S, Sosa J. Predictors of survival in sinonasal adenocarcinoma. J Neurol Surg B Skull Base. 2015;76:208-213. doi:https://doi.org/10.1055/s-0034-1543995
- Bhayani M, Yilmaz T, Sweeney A. Sinonasal adenocarcinoma: a 16-year experience at a single institution. Head Neck. 2014;36:1490-1496. doi:https://doi.org/10.1002/HED.23485
Downloads
License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright
Copyright (c) 2026 Società Italiana di Otorinolaringoiatria e chirurgia cervico facciale
How to Cite
- Abstract viewed - 271 times
- PDF downloaded - 54 times




